Glucose is probably the most discussed molecule in our body (often under its other moniker, blood sugar) and several Nobel prizes have been won studying it. For good reason: It’s a primary source of energy that powers our bodies at the cellular level (there are others, like ketone bodies, derived from fat). All living organisms can use glucose for energy.
Glucose is a simple carbohydrate—a monosaccharide, which means it is a single sugar. (Sucrose, or table sugar, is a disaccharide composed of glucose and fructose.) Its name comes from “gleukos” (Greek for “sweet wine”). The ending “ose” now tells us that something is chemically a sugar. And although this simple sugar is just one molecule, it plays a vital role in the functioning of almost all the cells in our body.
Our body needs glucose, so it has a complex system for making sure we don’t have too much or too little, either of which can cause a range of health problems. Too much, and you can wind up with issues ranging from obesity to Alzheimer’s dementia to blindness. Too little, and you could experience confusion, weakness, or even a coma. But this homeostatic system can be overwhelmed if we don’t make diet and lifestyle choices that help our bodies maintain stable glucose levels.
Here’s how your body processes glucose, what happens when that system malfunctions, and how you can maintain healthy glucose levels.
Learn more:
How glucose moves through your body
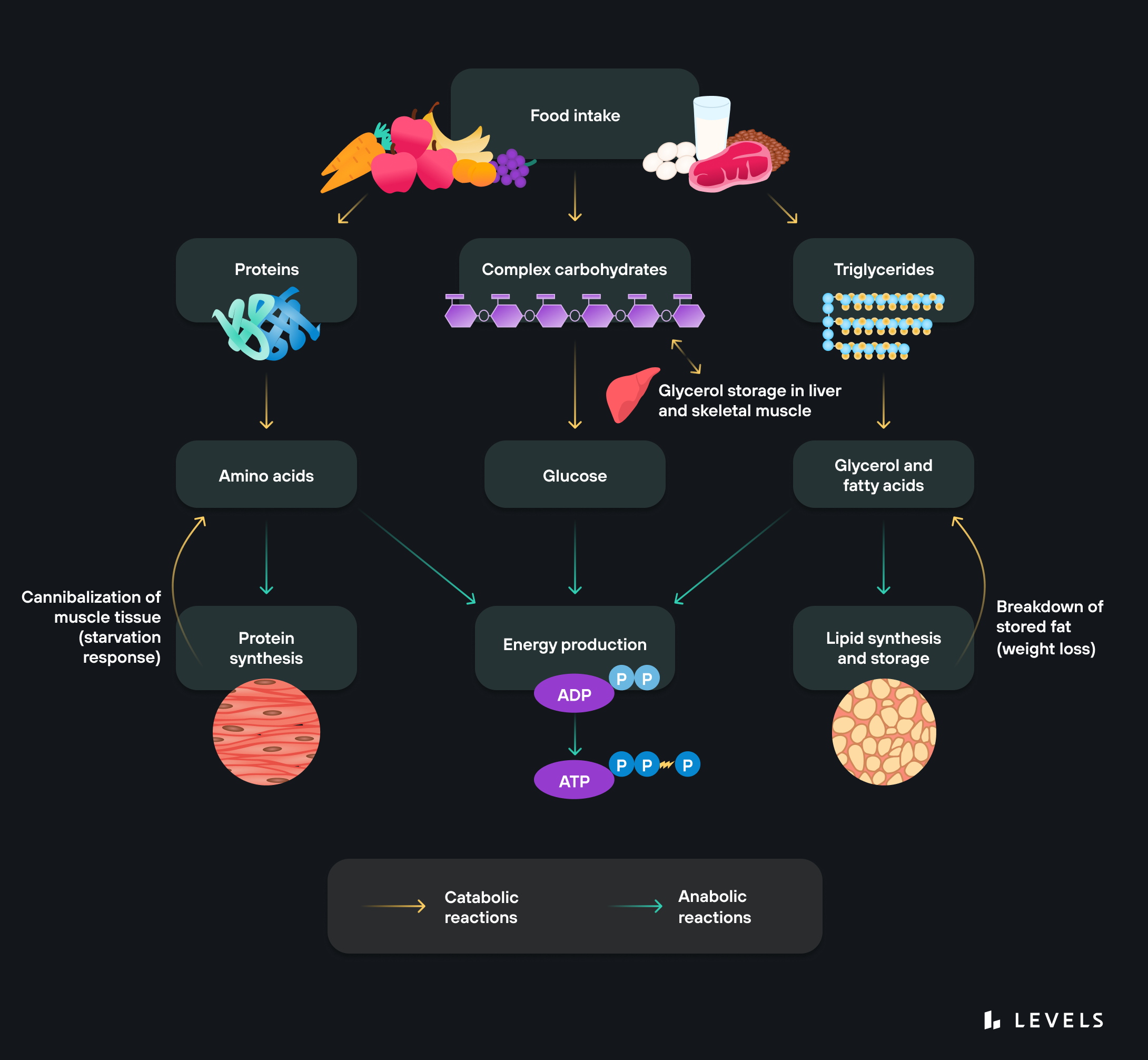
Our bodies can make glucose (more on that below), but most of what we use, we get from eating. Glucose is present in all plants and animals, usually in more complex carbohydrate forms like lactose, sucrose, and starch. So what’s a carbohydrate? Carbohydrates are a combination of hydrogen, carbon, and oxygen and one of the three macronutrients (along with fat and protein, or amino acids) your body uses for energy. They can be simple (meaning they have fewer than three sugars and are absorbed and processed quickly, typically leading to a blood sugar spike) or complex (more sugars, more extended processing period).
Here’s a quick look at how glucose goes from that donut you eat to the energy that powers your body’s cells.
It may help to start with a basic view of the path glucose travels: from your mouth down into your small intestine; then out into the bloodstream, where it takes a quick trip through the liver and moves through your body via your blood; it leaks out of the capillaries, into the interstitial fluid around the cells, and finally through the cell membrane and into the cell.
Sensing: As soon as you start eating, receptors on your tongue and in your gut and pancreas sense the presence of carbohydrates. This sets off a chain reaction of events to tell the body to process and use this fuel.
Signaling: As soon as the body detects glucose, a complex set of signaling instructions is triggered. For example, glucagon-like peptide-1 (GLP-1) tells beta cells in the pancreas to produce and release more insulin, which helps direct the glucose to cells and helps the cells absorb it.
Breakdown: Enzymes throughout your digestive system, including your saliva, pancreas, and small intestines, begin breaking down polysaccharides into glucose. (The enzyme at work depends on the form of the carbohydrate—amylase works on starches, lactase works on lactose, sucrase on sucrose, and so on.)
Transport: To get into the blood, glucose uses several different transporter molecules like SGLT and the GLUT family of proteins, which move glucose into various tissues. From the small intestine, it moves across the epithelial cells lining the intestine and then into the blood vessels. Once in the bloodstream, the glucose goes through the liver, where it either gets turned into glycogen and stored for later use or continues into circulation.
Absorption: Once glucose gets to the cells, insulin (and other hormones) help cells absorb it. In muscle and fat cells, for instance, insulin binds to cell surface receptors and causes GLUT4 channels to come to the cell membrane and ferry the glucose molecules inside. Other transporters work on other cells.
Oxidation: Once in the cell, mitochondria produce ATP, the energy currency of our cells, through the breakdown of glucose in the presence of oxygen. This process is known as aerobic or cellular respiration.
Storage: Not all ingested glucose gets used immediately—some goes into storage (a process also directed by insulin). Glucose can be converted to glycogen and stored in the liver or muscles, or into triglycerides and stored in fat tissue. Turning stored glycogen into glucose for energy is called glycogenolysis. Insulin directs glucose into storage, glucagon controls its release from storage.
Production: Our body can make glucose if it needs more than we’re ingesting. Glucose production happens primarily in the liver. It’s called gluconeogenesis and is roughly the reverse of glycolysis, the process of breaking down glucose. When glucose is low, because you’re fasting or have taken in few carbohydrates, your body will turn to gluconeogenesis to fuel certain organs, like the brain (which consumes around 60% of your blood glucose in the sedentary and fasted state and stores minimal glycogen).
Learn more:
Why do we talk about blood glucose?
Glucose is present just about everywhere in your body, but the place we most often measure it is in the blood. We talk about the concentration rather than the absolute amount, measured in milligrams (mg) of glucose per deciliter (dL) of blood. So how much do we have? As vital as this molecule is, not that much: Under normal conditions, the average person has around 4g of circulating glucose, about a teaspoon. (That doesn’t include glycogen, or stored glucose: We have around 100g of that in the liver and 400g in the muscles.)
“Close to 40% of all U.S. adults are known to have insulin resistance.”
We talk about “circulating” glucose, but a particular glucose molecule is never in the bloodstream very long, as it is continuously being delivered to cells and replenished. An incredibly intricate set of physiological processes has to work together to maintain that homeostatic level: between about 70 mg/dL and 100 mg/dL (more on those ideal levels below). It’s a constant tug of war between insulin and glucagon to either remove glucose from the blood (insulin) or put more into the blood from storage (glucagon).
Why do we care about blood glucose levels?
Glucose is one of the rare molecules in our body where we can and should monitor our current levels. For example, we don’t talk about our levels of amino acids or triglycerides nearly as much. That’s for a few reasons:
- Compared to most other metabolites (molecules involved in metabolism), glucose is easier to measure at home with simple devices like an over-the-counter finger-stick glucometer or a continuous glucose monitor (CGM).
- We have a lot of control over our glucose levels through our food and lifestyle decisions. Our blood sugar levels change throughout the day based in large part (though not entirely) on things we’re doing. If I decide to eat a giant bowl of sugary cereal, I can flood my body with glucose. That immediate cause and effect are less apparent with other metabolites.
- We can feel the effects of glucose pretty acutely. If we have high blood sugar, or hyperglycemia, even for a short time, we could feel jittery or excited. Low blood sugar (hypoglycemia) can cause fatigue, lethargy, or brain fog. Hypoglycemia can result from either a crash after a hyperglycemic spike or other causes, such as fasting.
The risks of high glucose levels
For most people, hyperglycemia—both reactive (short-term) and chronic (long-term)—is the more dangerous health risk.
Consistently high blood glucose is problematic not only because it can make you feel “off” but also because it triggers a flood of another hormone, insulin. Produced by beta cells in your pancreas, insulin is a crucial hormone that affects every cell in the body. It has several effects, but the most well-known is stimulating muscle and fat cells to take up sugar from the bloodstream to be used for energy or stored. (It has many other jobs that have nothing to do with nutrients, depending on the cell.)
However, if insulin levels are consistently high—say, because glucose levels are consistently high—cells can actually start to become “numb” to insulin’s effects, a condition called insulin resistance. That means your body needs more insulin to do the same work—this results in more circulating insulin and glucose.

What causes insulin resistance?
Insulin resistance is a shockingly common condition spurred on by diet and lifestyle. Here are the processes in your body that lead to this dangerous state.
Read the ArticleInsulin resistance is perhaps the most common health concern in the U.S. and can factor in nearly all chronic conditions, including heart disease, diabetes, infertility, and Alzheimer’s disease. Insulin blocks our body’s ability to burn fat for energy, so high levels are a critical factor in weight gain, and difficulty losing weight, which is highly relevant in the face of 72% of Americans being overweight or obese. Close to 40% of all U.S. adults are known to have it.
Chronically high glucose has other adverse effects beyond insulin. Excess glucose in the bloodstream can lead to chronic inflammation (the overactivation of the body’s immune response), oxidative stress (an overabundance of damaging free radicals in the body), and glycation (glucose molecules attaching to red blood cells). Too much glycation can produce harmful compounds called advanced glycation end products (AGEs) that can cause inflammation and lead to problems from wrinkles to Alzheimer’s disease.

Inflammation and glucose levels: How high blood sugar can turn a good system bad
Inflammation helps heal your body, but chronic inflammation can cause serious damage. Here’s how a healthy lifestyle—including a low-sugar diet—can help keep it in check.
Read the ArticleWhat affects blood sugar levels?
Diet
The biggest lever in controlling glucose levels is what we eat. Here are three basic rules for building a diet with stable blood sugar in mind:
- Avoid excess sugar. Put simply, consuming sugar is the fastest way to raise your blood sugar. Whether it’s added or natural, plain table sugar or the fanciest organic honey, sugars break down to glucose easily.
Eliminating sugar as best you can means reading labels, as there are dozens of types hiding in everything from ketchup to energy bars. But it’s not just added sugar—foods like fruit juices also pack a big glucose punch.
Note that natural sweeteners like stevia and monk fruit can be good alternatives; artificial sweeteners like Splenda and Equal have more mixed research and may hurt metabolic health over the long term. - Avoid “naked” carbs and limit overall carb intake. Our bodies convert all carbohydrates into glucose, regardless of whether they come from oatmeal, pasta, fruit, sweet potatoes, or a donut. When carbohydrates are the primary macronutrient in a meal and not balanced with fat or protein, we call them “naked,” and they may have a more significant impact on blood sugar.
Sugars like glucose, sucrose, or fructose are simple carbohydrates that are simplest to absorb into the blood. But more complex carbs in starchy foods like rice, cornmeal, or potatoes can also lead to a glucose rise, especially when eaten alone without fat, fiber, or protein to blunt their impact.
For example, if you eat an apple, pair it with nut butter or put it in a chia seed pudding (high fiber and healthy fats). If you have a sweet potato, add tahini, olive oil, flax seeds, or nuts, plus some protein.
If you see a high carbohydrate or “net carb” count on a label, maybe skip that food. (Net carbs are a measure of total carbs minus fiber and sugar alcohols; the relationship between net carbs and blood sugar rise is debated and depends on the type of fiber and sugar alcohols the food contains.) For unlabelled food, you can experiment with a CGM to learn how you react. - Avoid processed foods, including refined grains. The more processed the food, the more likely it is to cause a quick rise in glucose. These foods include anything made with refined grains and flour, like white bread, tortillas, crackers, or bagels, which your body will break down quickly, leading to a blood-sugar spike.
More processed and packaged foods are also more likely to have added sugars or other damaging additives and to have had much of their nutrient content stripped away.
Instead, try to eat:- Low-carbohydrate vegetables (e.g., greens, broccoli, cauliflower, zucchini, Brussels sprouts)
- Fiber and protein-rich foods like legumes (beans, lentils), nuts, and seeds
- Fats in their whole-food form like nuts, seeds, avocado, coconut, and olives
“How your body responds to food and its ability to get back to that set-point efficiently are critical markers for your blood-sugar health.”

12 Ways to lower blood sugar
Maintaining stable blood sugar is key to optimal metabolic health. Here are 12 strategies to improve glucose levels.
Read the ArticleExercise
Physical activity affects glucose levels for several reasons, including the fact that working muscle can need up to 10 times more glucose than sedentary muscle. This need spurs the liver to release glycogen to maintain that set level of glucose in the blood. Other hormonal processes dampen the amount of glucose muscles need. Muscle can absorb glucose without insulin, so glucose variations during exercise are less damaging than those from eating—they don’t trigger the same insulin flood.
Exercise is ultimately beneficial to glucose control. Multiple studies show that moderate activity for just 30 minutes, three times per week, can improve insulin resistance and glycemic control. Research has also found that many short bouts of movement throughout the day can be more beneficial than one long workout, so aim to be more active more frequently, even if it’s for just a few minutes.

Optimize your exercise performance by tracking glucose
Exercise, glucose, and metabolic flexibility: how monitoring glucose levels with CGM can help you improve fueling, endurance, performance, and recovery.
Read the ArticleStress
Stress and glucose have a two-way relationship. Studies show that perceived work stress alone can increase our circulating glucose levels. But raised glucose levels can also affect the body’s ability to deal with stress.
Stress elevates cortisol and epinephrine, which make muscle and liver cells acutely insulin resistant. This may be an evolutionary response designed to ensure that we have the energy to fight or flee, but when today’s stressors are more likely psychological, it’s a maladaptive response.
Fortunately, tools for lowering stress can also help lower glucose. Studies show that simple actions like twice-weekly meditations or 20-minute daily breathing can reduce fasting and post-meal glucose levels.
Sleep
The effect of sleep on glucose also involves cortisol. Under normal conditions, cortisol levels tend to be lower during the evening and early part of the night, so glucose levels usually stabilize around a lower range at night.
However, the body interprets poor sleep, or not enough sleep, as a stressor and releases excess cortisol, with the same adverse effects on glucose. Even a few nights of sleep deprivation can throw off your body’s ability to process glucose efficiently. An eight-year study of adult men in Japan found that those who had trouble falling and staying asleep had two to three times the risk of developing Type 2 diabetes.

How does sleep affect blood sugar?
Sleep quality and quantity have a profound effect on metabolic health. This article describes the mechanisms by which improving sleep hygiene can improve glucose control.
Read the ArticleHow to measure blood glucose levels and “normal” results
Testing blood sugar levels is part of the typical blood test in an annual physical as a screen for diabetes. You can also test your current glucose anytime at home with a $20 over-the-counter finger-stick glucose meter, where you draw a small drop of blood and place it on a test strip.
Fasting plasma glucose (FPG) test
Before your physical, your doctor will ask you to fast for eight hours. This is so they can measure your fasting plasma glucose, or the concentration of glucose in your blood. As you can imagine, if you ate a donut on your way to the appointment, your blood at that moment would show very high levels of blood sugar. Fasting gives us some approximation of your “typical” glucose concentration. Unfortunately, it’s still a single point in time, where, as we saw above, your level could be high or low depending on how well you slept the night before or how stressed you get at the doctor’s office.
So what results do you want to see? According to the American Diabetes Association (ADA), people fall into three categories depending on their fasting plasma glucose levels:
- Normal: Under 100 mg/dL
- Prediabetes: 100–125 mg/dL
- Diabetes: Higher than 125 mg/dL
It’s important to note that these are benchmarks—a fasting glucose level of 99 mg/dL is still less than ideal. And people with prediabetic or diabetic levels can work to bring their glucose back down to healthy ranges.
Post-meal glucose
While fasting glucose looks at your body’s natural set-point, how your body responds to food and its ability to get back to that set-point efficiently are critical markers for your blood-sugar health. Of course, we expect glucose to go up after a meal (though if we eat carefully, it doesn’t have to rise much). But if your body can’t return to that baseline quickly, it could indicate that insulin is not working well because you have insulin resistance. Measuring post-meal glucose gives us some insight into that functioning.
In a doctor’s office or lab setting, we measure this with an oral glucose tolerance test, giving people a drink with 75g of glucose and then test their blood two hours after. If glucose levels are less than 140 mg/dL at two hours after consuming the glucose drink, a patient is considered “normal.” One challenge with this is that it’s not a great simulation of how we usually get sugar into our bodies (unless you drink a lot of Kool-Aid). It’s also another single-point-in-time measurement.
According to the International Diabetes Federation (IDF), nondiabetic people should have a glucose level of no higher than 140 mg/dL after any meal, and glucose should return to pre-meal levels within 2-3 hours. Hyperglycemia is a level over 140 mg/dL 1–2 hours after eating.
We would argue these are pretty high targets. Based on data from studies of people without diabetes wearing CGMs, we suggest a post-meal glucose goal of less than 110 mg/dL, with no more than a 30 mg/dL increase from pre-meal levels.

What should your glucose levels be? Here’s the ultimate guide to healthy blood sugar ranges
Fasting blood sugar levels classify into 3 categories: normal, prediabetes, and diabetes. To be considered “normal,” fasting glucose must be under 100 mg/dl.
Read the ArticleA1c or HbA1c
Available only at your doctor’s office, this blood test measures the percentage of red blood cells with glucose attached to them. Unlike a fasting glucose test, which captures a moment in time, A1c shows your average glucose levels over the past three months (about how long glucose stays attached to hemoglobin, the Hb in the title). However, it’s still limited in that it doesn’t reflect daily glucose variability, which we know can affect health outcomes. Here’s a rough guide to the numbers:
- Normal: Below 5.7%
- Prediabetes: 5.7% to 6.4%
- Diabetes: 6.5% or above
Glycemic variability
Knowing what we do about the damage caused by high and low blood sugar, the limitations of single-point measurements become clearer. Someone regularly spiking and crashing their glucose levels could still show a “normal” A1c or FPG. Glycemic variability is a measure of the up-and-down swings in glucose throughout the day.
This is only possible to measure with a continuous glucose monitor, which takes readings from the interstitial fluid under your skin throughout the day.
Glucose and Diabetes
Diabetes describes a condition where the body’s glucoregulatory system doesn’t work as intended. There are two primary types of diabetes:
Type 1 diabetes is when your body doesn’t make enough insulin to regulate glucose in your blood. It’s thought to be an autoimmune disease in which your body attacks the pancreas’ beta cells. It often appears in children or young adults but can develop at any age. People with T1D have to closely watch their glucose levels either with a continuous glucose monitor or by doing finger stick tests up to a dozen times a day. They have to augment their body’s insulin with either injections or an insulin pump.
Type 2 diabetes is much more common: About 1 in 10 Americans have diabetes, and 90–95% of them have Type 2. Unlike Type 1, Type 2 is often preventable. It’s a form of extreme insulin resistance, where chronically elevated insulin levels cause cells to “numb” to the hormone’s effects, so glucose levels can get dangerously high if not carefully regulated.
Prediabetes refers to glucose levels that are high but not yet at the level of Type 2 diabetes. A prediabetes diagnosis is a warning, and like Type 2, we can reverse it with diet and lifestyle changes that bring glucose levels back down.
Gestational diabetes occurs during pregnancy and is usually a temporary condition, although it can increase the risk of developing Type 2 diabetes later. Pregnancy naturally increases insulin resistance, as the placenta needs to store more glycogen to help the fetus grow (insulin is a “building” hormone as opposed to a “breaking down” hormone). But if the insulin resistance begins to affect the body’s ability to process glucose, it can lead to elevated glucose levels: gestational diabetes.
Summary
Glucose is one of the most essential molecules in your body, providing most of the energy your cells need to function. Because it affects every cell in our bodies, disruption in our glucose levels can lead to problems that manifest just about anywhere, from the skin to the brain to organs to our nervous system. It can also disrupt hormones, like insulin, leading to more conditions. Fortunately, glucose levels are largely in our control. Through consistency with a healthy, thoughtful diet and positive lifestyle decisions, we can keep our glucose stable and our bodies healthier for it.