Humans are complex organisms with unique genetic, biochemical, and microbial blueprints. The reality is that every bite we consume impacts whether our bodies are moving toward optimal health or dysfunction. While a standard guidebook for living would be nice, each person must determine the unique diet that supports their body’s highest level of functioning. Fortunately, continuous glucose monitoring (CGM) can be a powerful tool in this pursuit. CGM can provide data and feedback to help determine how daily choices affect glucose levels in real time. We can finally write our own guidebooks, linking the meals we eat with metabolic health with actionable information that can inform our food choices. We understand, it’s tough out there. We’re surrounded by what feels like unlimited sources of food, plenty of which claim to be “healthy.” But despite this abundance, our country is sicker than ever. Recent research shows that in 2018, 80% of consumers found conflicting information about food and nutrition, and 59% said that makes them doubt their choices. The beauty of objective data is that it cuts through the noise.
What’s an Optimal Diet? Glucose Levels Can Help You Find Out
Glucose levels and how they change over the long and short term have a great deal to do with health and well-being. Diet is the most significant determinant of our blood sugar. Chronically elevated levels and post-meal spikes can lead to serious dysfunction, including increased risk for Type 2 diabetes, cardiovascular disease, stroke, liver cirrhosis, obesity, death from cancer, and more. Healthy regulation of glucose is a fundamental building block of an optimal diet.

12 Ways to lower blood sugar
Maintaining stable blood sugar is key to optimal metabolic health. Here are 12 strategies to improve glucose levels.
Read the ArticleIn the past, there have been a few tools to help understand how food affects glucose levels. A glycemic index chart could offer rough estimates of a food’s predicted glucose impact for the general population. Daily finger sticks to spot-check glucose levels could provide more personalized information. CGM changed all this. This powerful tool can track glucose trends 24 hours a day, not only in response to diet but for lifestyle behaviors, including exercise, sleep, and many others that affect glucose. A diet that optimizes glucose levels should have three main goals:
- Minimizing spikes after meals
- Maintaining levels in a relatively narrow and healthy range
- Keeping fasting glucose (measured after consuming no calories for at least 8 hours) in a range that carries the lowest risk
Minimize Glucose Spikes After a Meal
The term postprandial hyperglycemia refers to larger-than-average elevations, or spikes, in glucose levels after eating a meal. Excessive spikes are dangerous for many reasons. They’re a risk factor for developing Type 2 diabetes, cardiovascular disease, thickened carotid artery walls, liver disease, obesity, stroke, retinopathy, renal failure, cognitive dysfunction, cancer, and mortality. The mechanisms linking glucose spikes with chronic disease may include oxidative stress and inflammation, which can contribute to insulin resistance. Insulin resistance is when the body’s cells become less responsive—or “numb”—to signals from insulin, a hormone that allows cells to take up glucose. Insulin resistance can be the first step toward diabetes.

What is insulin resistance? (with video)
When a cell loses its responsiveness to insulin, which can happen due to various conditions, it becomes insulin resistant.
Read the ArticleScientists believe it proceeds like this: Insulin resistance leads to lower post-meal blood glucose control, followed by elevated fasting glucose levels, which leads to sustained high glucose levels over time. As a point of reference from the UK’s National Institute for Clinical Excellence, healthy people should rarely exceed 140 mg/dL after a meal, and glucose should revert to pre-meal levels within two to three hours. However, studies of non-diabetic populations wearing CGMs suggest that we may benefit from keeping an even tighter range after meals. One study showed that healthy young adults spent about 80% of the time at glucose levels under 100 mg/dL and less than 1% over 140 mg/dL. Another study showed that healthy people spend 94.4% of the time at glucose levels under 120 mg/dL and, again, less than 1% above 140 mg/dL. So, aiming to stay below 140 mg/dL after meals is likely too lenient of a benchmark for optimal health. A healthy person should more likely keep their glucose below 100 mg/dL for most of the day and rarely exceed 120 mg/dL.
“Glycemic variability increases as people move along the continuum from normal glucose regulation toward diabetes.”
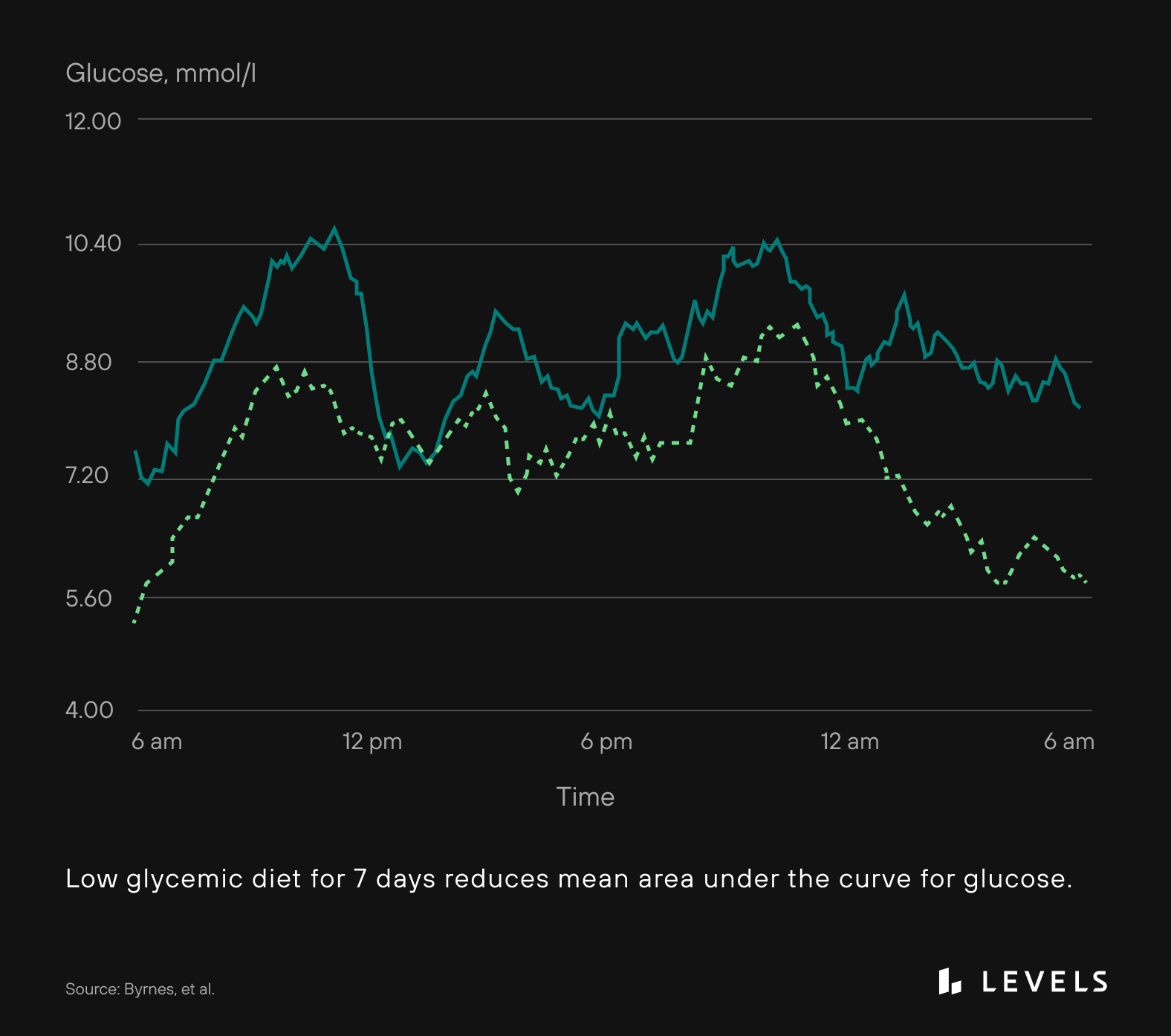
The implications are clear. When seen through the lens of glucose levels, an optimal diet should focus on foods that minimize post-meal spikes. This kind of diet can reduce the risk of developing diabetes, heart disease, and some cancers. They can also improve insulin sensitivity and cholesterol profiles. The concept of the glycemic index (GI) has pretty simple parameters: It describes the rise in glucose levels observed after the intake of 50 grams of carbohydrates of a specific food. Under this paradigm, a food with a higher glycemic index raises glucose levels more than a food with a lower index. However, the GI has limited application in the real world because very few people eat 50 grams of a particular carbohydrate at a time. Despite this, some studies find that a diet’s glycemic index is a good predictor of blood sugar fluctuations and that low glycemic index diets can reduce post-meal glucose elevations (see Figure 1).
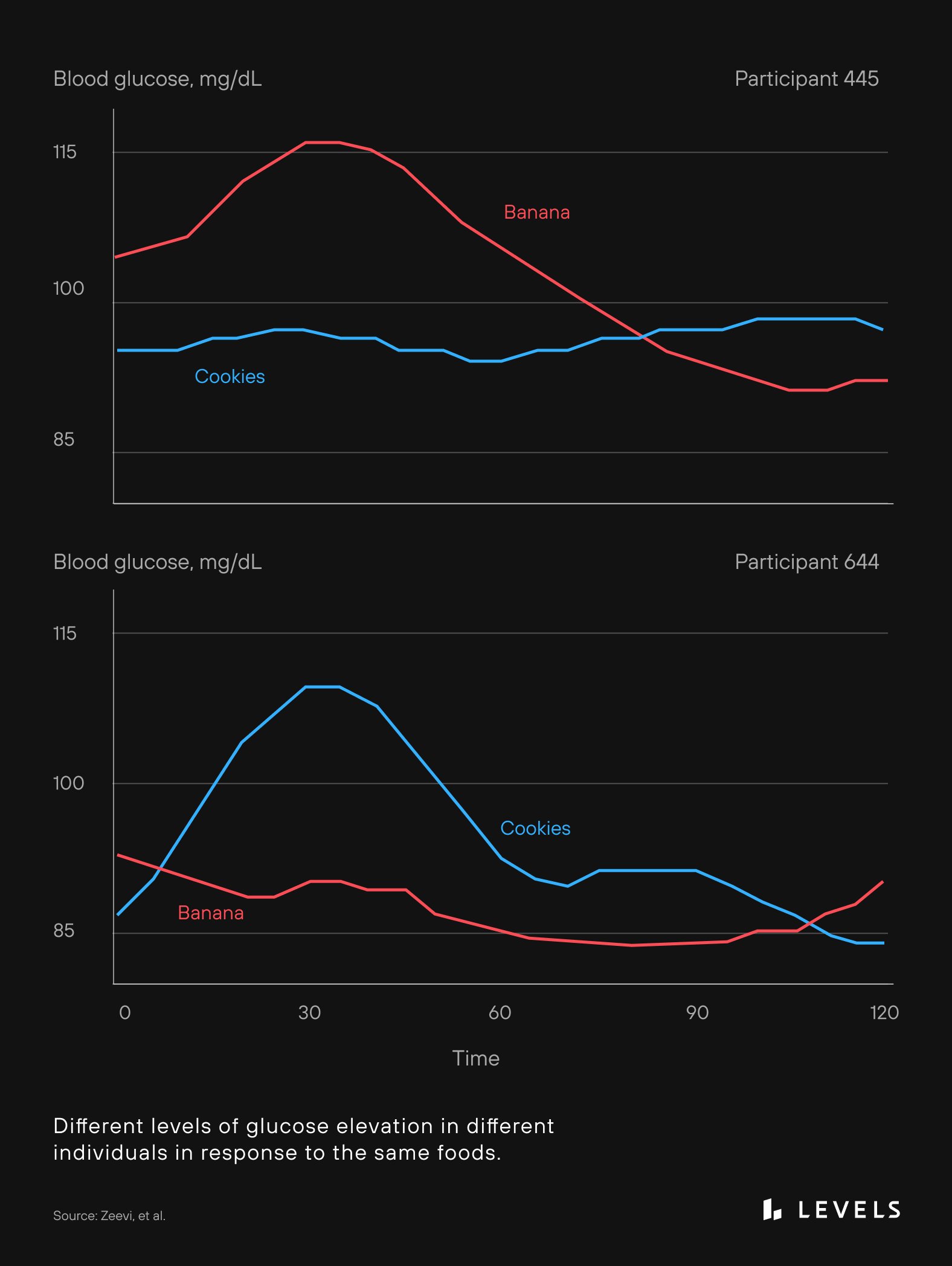
However, other research shows that two peoples’ glucose responses can vary significantly, even after eating the same food. This introduces some uncertainty into the idea of standardized scales to predict glycemic response to foods.
Traditional metrics support low-glycemic diets in the general population, but they may not accurately predict glycemic response on an individual level. It’s more likely that personal genetics, lifestyle, physical activity, body type, and microbiome also play an essential role in reducing glucose spikes after a meal.
Limit Variability in Glucose Levels
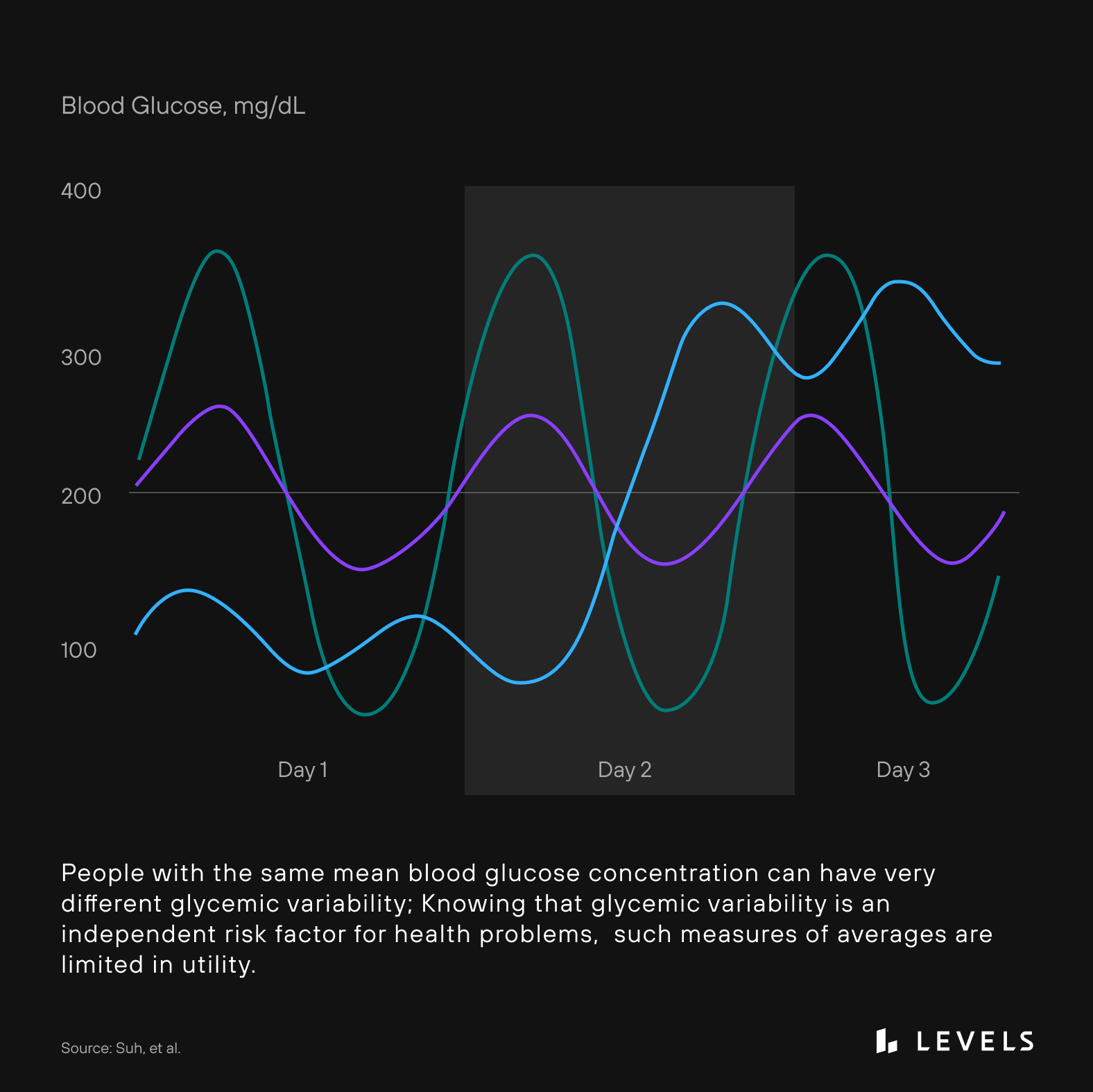
Glycemic variability refers to large swings in glucose levels. Also known as glycemic excursions, these are potentially more harmful than sustained high glucose levels alone. It’s thought that excessive peaks and dips in glucose can lead to tissue-damaging metabolic byproducts such as free radicals, damage to blood vessels, damage to the nervous system, inflammation, and activation of the stress hormone cascade (called sympathetic nervous system activation). Glycemic variability increases as people move along the continuum from normal glucose regulation toward diabetes. As a person becomes more insulin resistant, they’ll also tend to show more variability in glucose levels—fasting and post-meal. Simply put, poor glycemic regulation isn’t just about having higher glucose levels but also about increased variability.
The average height of glucose spikes and dips—known as “mean amplitude of glycemic excursions” or MAGE—is lowest in those without diabetes or obesity. A normal range for nondiabetic, non-obese people is between 26-28 mg/dL. In contrast, a non-diabetic, morbidly obese individual displays a MAGE value of 48.6 mg/dL. MAGE increases in those who are prediabetic or obese are higher still in those with stable diabetes and are highest in uncontrolled diabetes. Clearly, for best health, we want to eat in a way that produces minimal glycemic excursions. Unfortunately, a hemoglobin a1c test (a standard test that measures approximate average glucose over three months) doesn’t take these spikes and dips into account, so it may miss this critical, independent risk factor for diabetes and many chronic diseases (see Figure 3).
Fasting Glucose and “Normal” Ranges
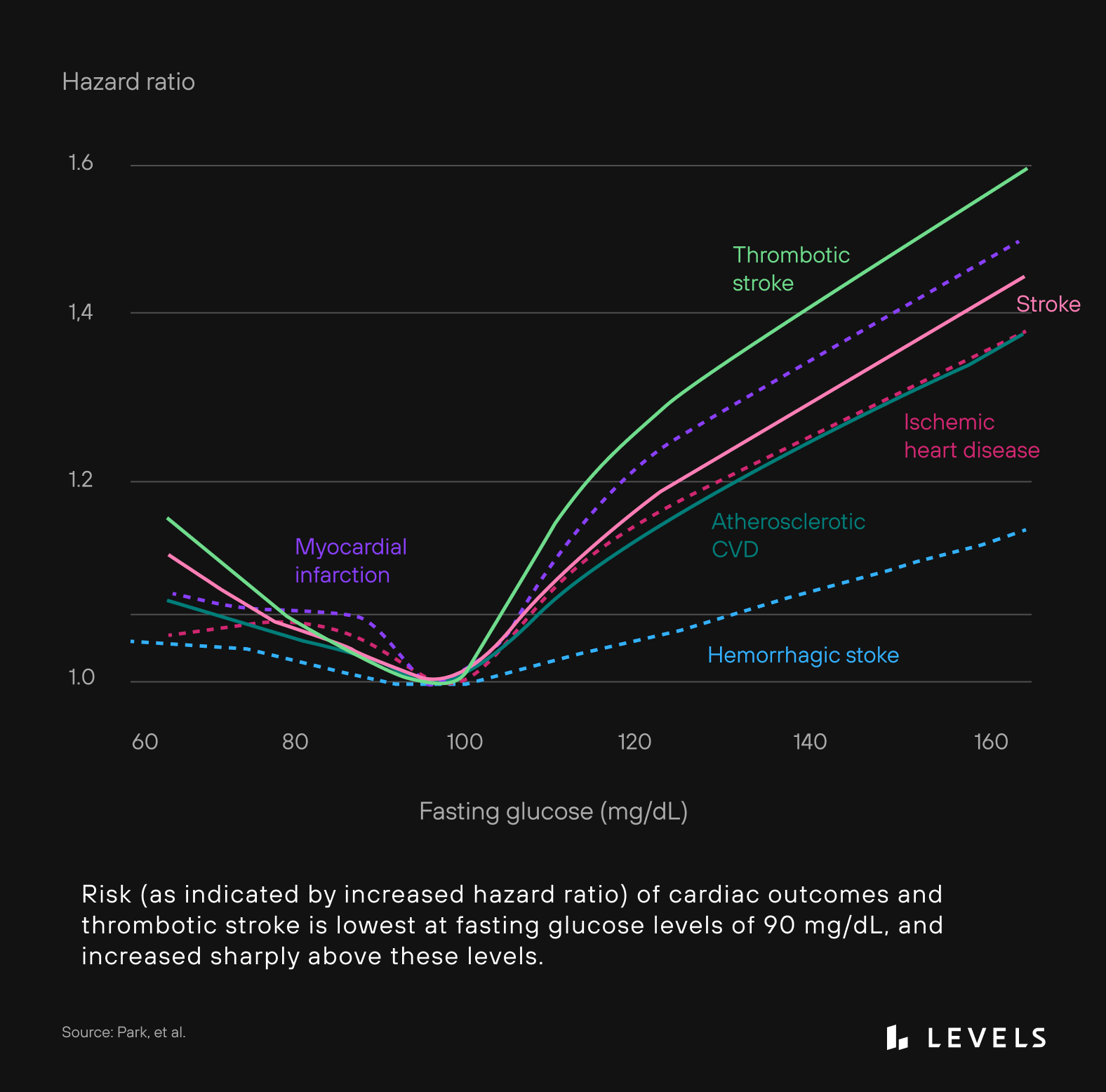
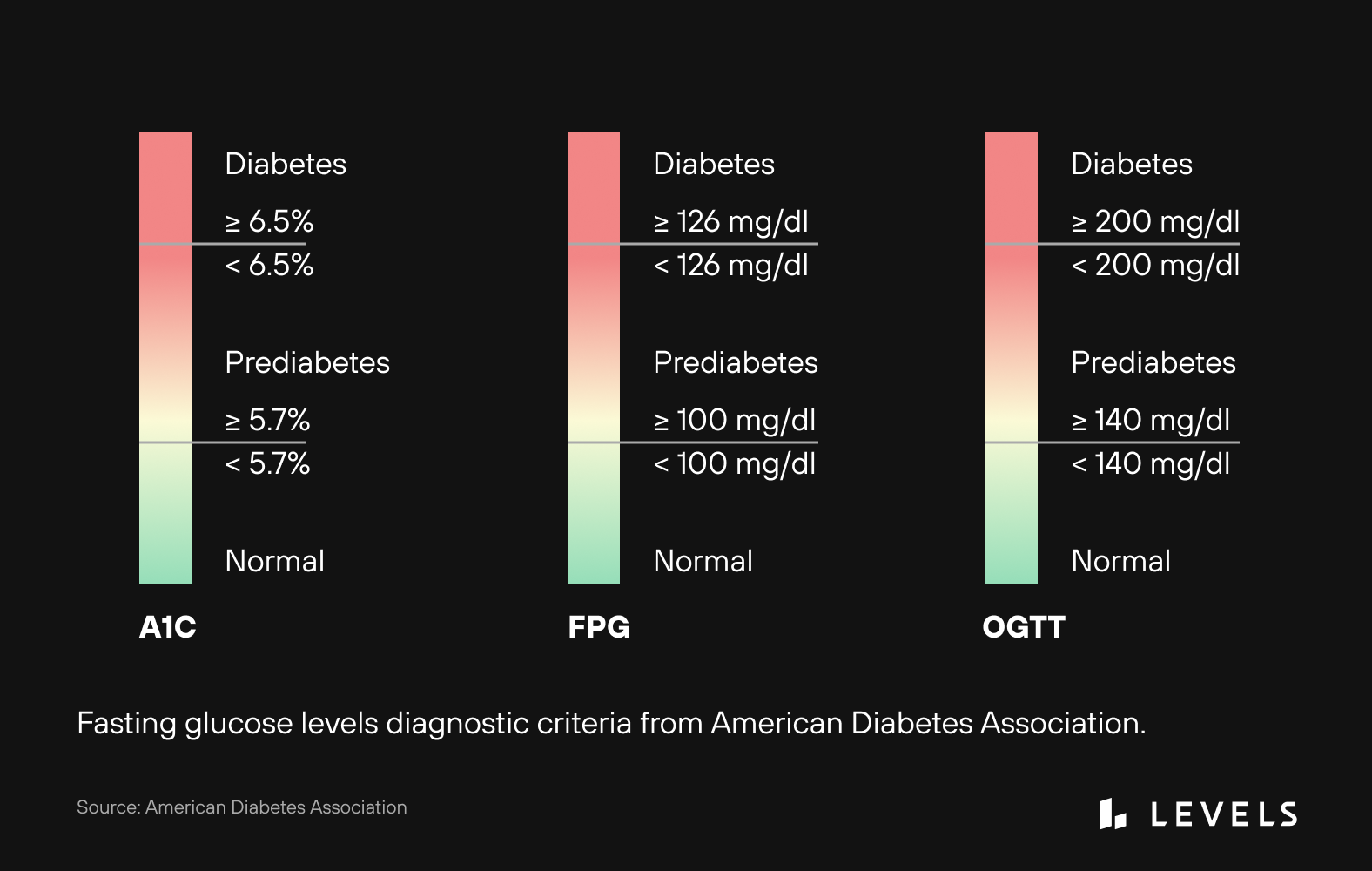
“Fasting glucose” measures glucose levels after consuming no calories for at least 8 hours. A high number is a strong predictor of developing diabetes down the road and may even be a risk factor for heart disease and stroke, among other health problems, even when this level is in the non-diabetic “normal” range (see Figure 4). The American Diabetes Association places fasting plasma glucose levels into three categories: “normal” (less than 100 mg/dL), “prediabetes” (100-125 mg/dL), and “diabetes” (more than 126 mg/dL) (see Figure 5). Up to 70% of people at the prediabetes level will eventually develop diabetes, and approximately 80% aren’t aware of their condition!

What should your glucose levels be? Here’s the ultimate guide to healthy blood sugar ranges
Fasting blood sugar levels classify into 3 categories: normal, prediabetes, and diabetes. To be considered “normal,” fasting glucose must be under 100 mg/dl.
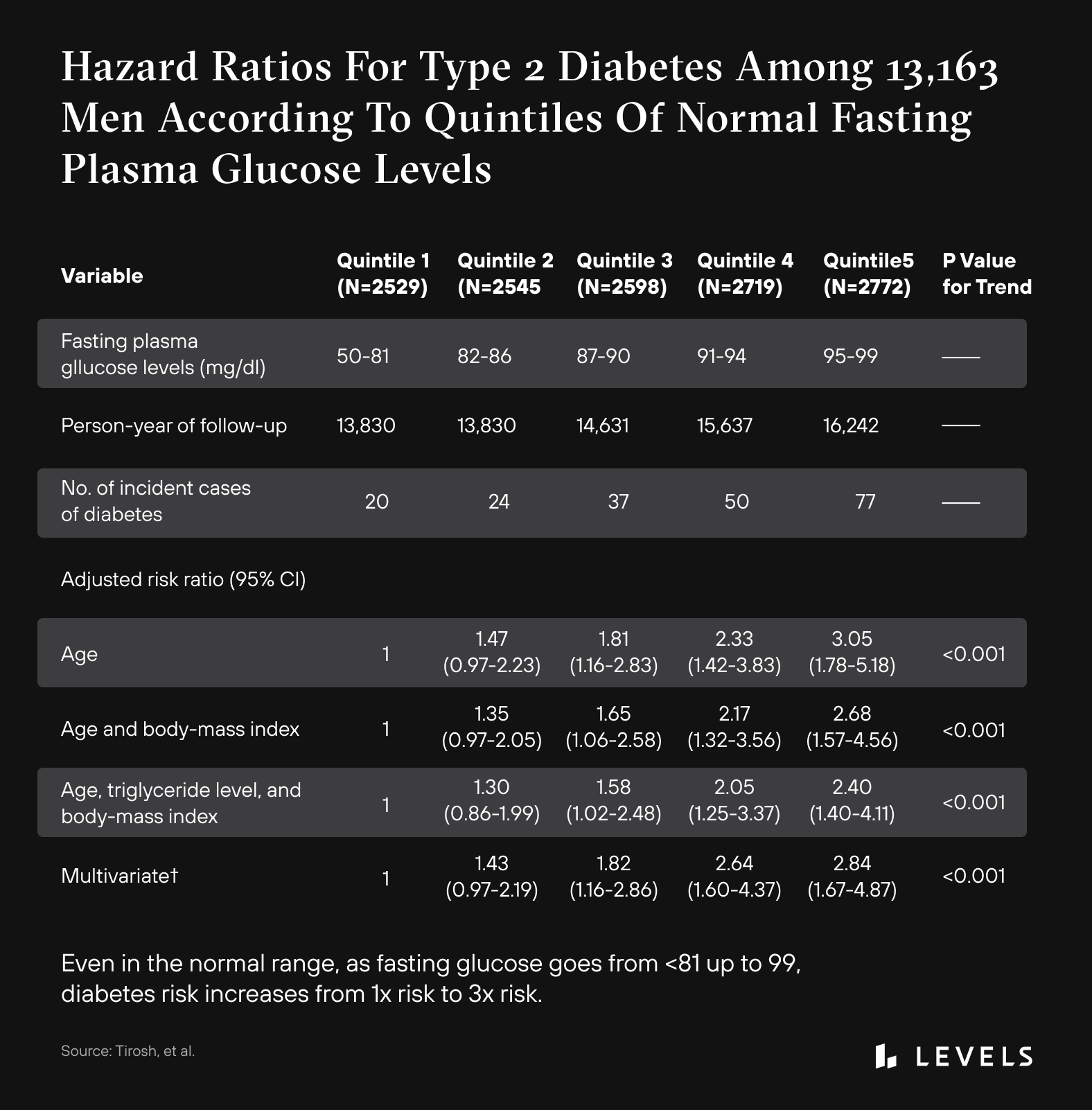
Read the ArticleAdditional research indicates that even people in the high “normal” range are at an increased risk of developing diabetes. Research in children has shown that having a fasting glucose measurement of 86-99 mg/dL, while still in the “normal” range, increases the risk of developing prediabetes and Type 2 diabetes more than two-fold when compared with children with a fasting glucose less than 86 mg/dL. A large study in adults showed that as fasting glucose increases from under 81 up to 99 mg/dL, there is an increase in the risk of developing diabetes by as much as three times, despite all of these people meeting the criteria for “normal” fasting glucose. What’s more, there is a sharp increase in cardiovascular disease risk, heart attacks, and thrombotic stroke as fasting glucose increases. This increase in risk begins at ~90 mg/dL fasting glucose, well below the “normal” fasting glucose level cut-off of 100 mg/dL. The idea of a fasting glucose of under 100 mg/dL being a threshold for complete safety and risk avoidance is flawed. Physiology is a spectrum, and it is up to every person to stay vigilant and aware of where they fall on this spectrum. Unfortunately, traditional tools for assessing glucose don’t give us this level of granularity. Tools like CGM can help, and you can improve glucose levels with dietary and lifestyle changes.
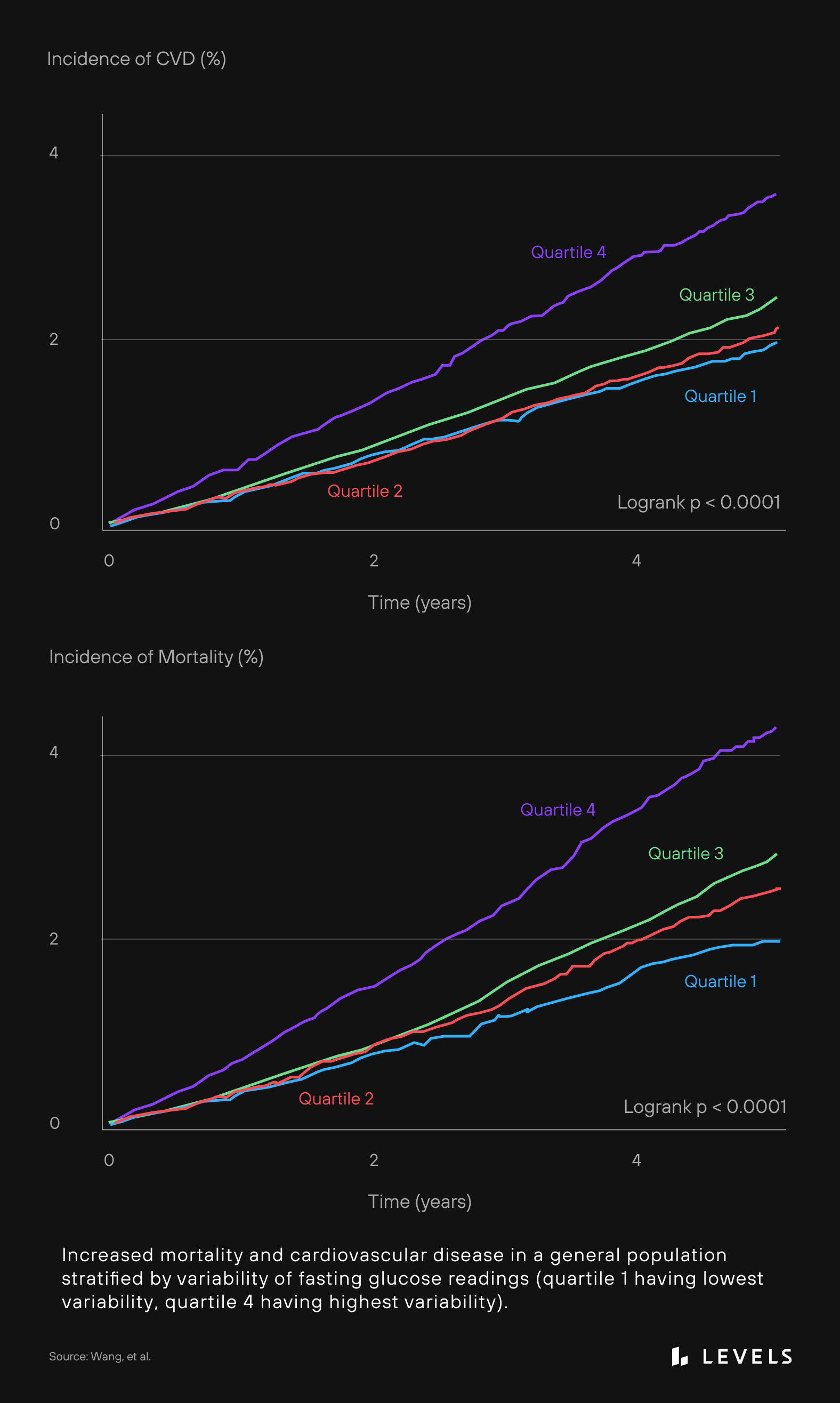
Variability between readings may also be a risk factor. Fasting glucose levels that “bounce around” from test to test appear to correlate with increased heart disease and mortality risk. Research shows that those with the least variability between tests have a lower risk of health problems (see Figure 7).
An optimal diet should consider the diet’s effect on keeping fasting glucose on the lower end of the normal range and minimizing variability between test-to-test fasting levels. The good news is that CGM can be a powerful tool to monitor glucose and determine which foods lead to better or worse glycemic function. Consistent use of these devices can support lifestyle changes that lower fasting blood glucose. And these lifestyle changes can work dramatically—even for those who meet the criteria for prediabetes, they can reduce the risk of progressing to diabetes by up to 70%.
Optimal Diet: There’s No “One Size Fits All”
The data makes it clear: No one-size-fits-all diet will optimize glucose. Neither a food’s carbohydrate content nor glycemic load/index can predict a person’s exact response to a real-life meal. Standardized scoring systems like GI aren’t personalized enough (see Figure 2). Moreover, these metrics fail to account for genetics, weight, sleep quality, stress levels, gut microbiome, insulin sensitivity, and food combinations—all of which affect glycemic response. So, what’s the best way to determine an optimal diet? It’s simple, but it requires data and analytics. We know that an optimal diet minimizes post-meal glucose spikes, reduces glycemic variability, and maintains fasting blood glucose in an optimal range. CGM can aid in achieving these aims. Using CGM (especially coupled with software to interpret the data), you can track post-meal spikes, glycemic variability, and fasting glucose in a way that’s accurate, individualized, applicable to real life, and actionable. Reading nutrition labels isn’t enough. CGM can offer enhanced control and power to pursue optimal health and well-being and take the guesswork out of your diet.