This newsletter was reviewed by board-certified orthopedic surgeon Dr. Howard Luks, who can be found on Twitter and his blog. Stay tuned for our Whole New Level episode with him, coming soon.
In June, I was doing back squats in a CrossFit class, and the next day I had painful right knee swelling. An MRI showed I’d torn 1.4 centimeters of cartilage off my femoral condyle. My orthopedic surgeon recommended a complex surgical procedure: The first stage would harvest cartilage and microfracture the bone to promote scarring. They would grow the cartilage in a lab and then do a second surgery to re-implant my cartilage into my knee. Undergoing this two-stage surgery (called a MACI procedure) would mean I couldn’t walk for five months post-op!
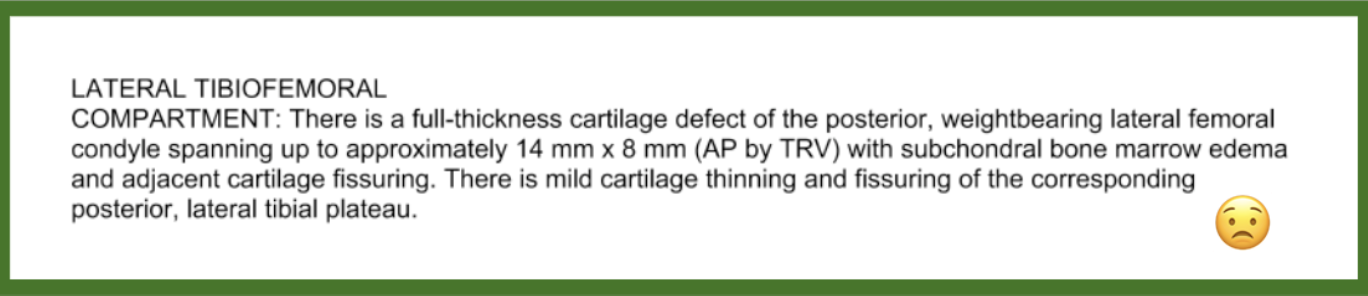
My diagnosis.
The rationale for this procedure was to reduce the risk of possible development of future arthritis. Arthritis is pain and inflammation in a joint, and since cartilage cushions the lining of most joints, a cartilage tear can theoretically contribute to inflammation and development of arthritis. However, my pain wasn’t too intense. So—against the surgeon’s strong recommendation (telling me, “If this were me, I’d have the MACI”)—I decided to hold off on surgery. I consulted two additional board-certified orthopedic surgeons who have expertise in regenerative and holistic therapies. Both strongly encouraged me to wait on any procedure, do as much physical activity as I could tolerate, maintain a healthy weight, and continue a healthy, anti-inflammatory diet and lifestyle to support healing and regeneration of my joint.
I took that advice, and now, four months later, I am fully pain-free. My specific recovery plan took the form of regular cold plunging, a whole foods anti-inflammatory diet, lots of movement and gradually increasing resistance training, supportive supplements (like omega-3s, curcumin, urolithin A, and others [more below]), and mindfulness practices. It took a few months for the pain, swelling, and clicking sensation with every step to subside, but now I am squatting, jumping, jogging, biking, hiking, and performing 220-pound leg presses without symptoms. Since the injury, I’ve backpacked 60 miles in Wyoming with a 40-pound pack, biked 30 miles with 3,000 feet of elevation gain, ran one of my fastest miles ever, and done a handful of local 10- to 15-mile day hikes with 2,000+ feet of elevation gain. I shudder to think how life would have looked had I undergone the surgery and been off my feet for nearly half a year.
This experience and outcome with a conservative approach to therapy has caused me to reflect on a dominant mentality in Western medicine: We are laser-focused on invasive, “hammer” interventions that permanently change structure (i.e., surgery) or temporarily impact symptoms (i.e., opioids). But what these aggressive interventions lack is an emphasis on creating conditions in the body that support its programmed healing and regenerative capacities, many of which are rooted in metabolic optimization. A body that can’t make energy properly (one that is experiencing metabolic dysfunction) is less resilient and can’t heal optimally.
My journey with my knee injury got me digging into the connections between joint health and metabolic health. The information is extremely empowering; it reminds us that we are not bystander victims to an inevitable fate of pain, reduced mobility, dependence, and suffering. Anytime I can integrate actions that support my body’s own healing, I want to implement them. I hope the healthcare system follows suit. In this newsletter, I cover:
- How arthritis/osteoarthritis (OA) is not just a structural issue but a biological issue tied to metabolic health.
- The mechanisms of how metabolic dysfunction and musculoskeletal dysfunction are linked.
- An exploration of how to expand our thinking about surgery for some musculoskeletal issues.
- How dietary changes and even some supplements can help the musculoskeletal system and joints heal.
- How our mind-body connection relates to pain and health outcomes.
Joint health and metabolic health are connected
Our joints are a framework of bones, cartilage, ligaments, and tendons. Cartilage cushions our bone ends from various forces, while ligaments provide joint stability. Tendons connect muscle to bone. These tissues work together to support us and help us move. Arthritis/osteoarthritis (OA), which affects more than 32.5 million adults in the U.S., is often called “degenerative” or “wear and tear” arthritis, but this does us all a disfavor. In fact, it may be better to think of OA as a metabolic disease where the tissue’s ability to make and use energy is impaired. The Venn diagram of metabolic issues and joint issues contains a huge overlap in mechanisms: things like chronic inflammation, blood flow problems, mitochondrial dysfunction, and more. Additionally, experiencing obesity—which is associated with low-grade chronic inflammation—puts someone at a five times higher risk for both metabolic syndrome and OA. It shouldn’t come as a surprise that Type 2 diabetes and OA are two of the most common chronic conditions we face today and they are deeply interlinked mechanistically. Here are five connections.
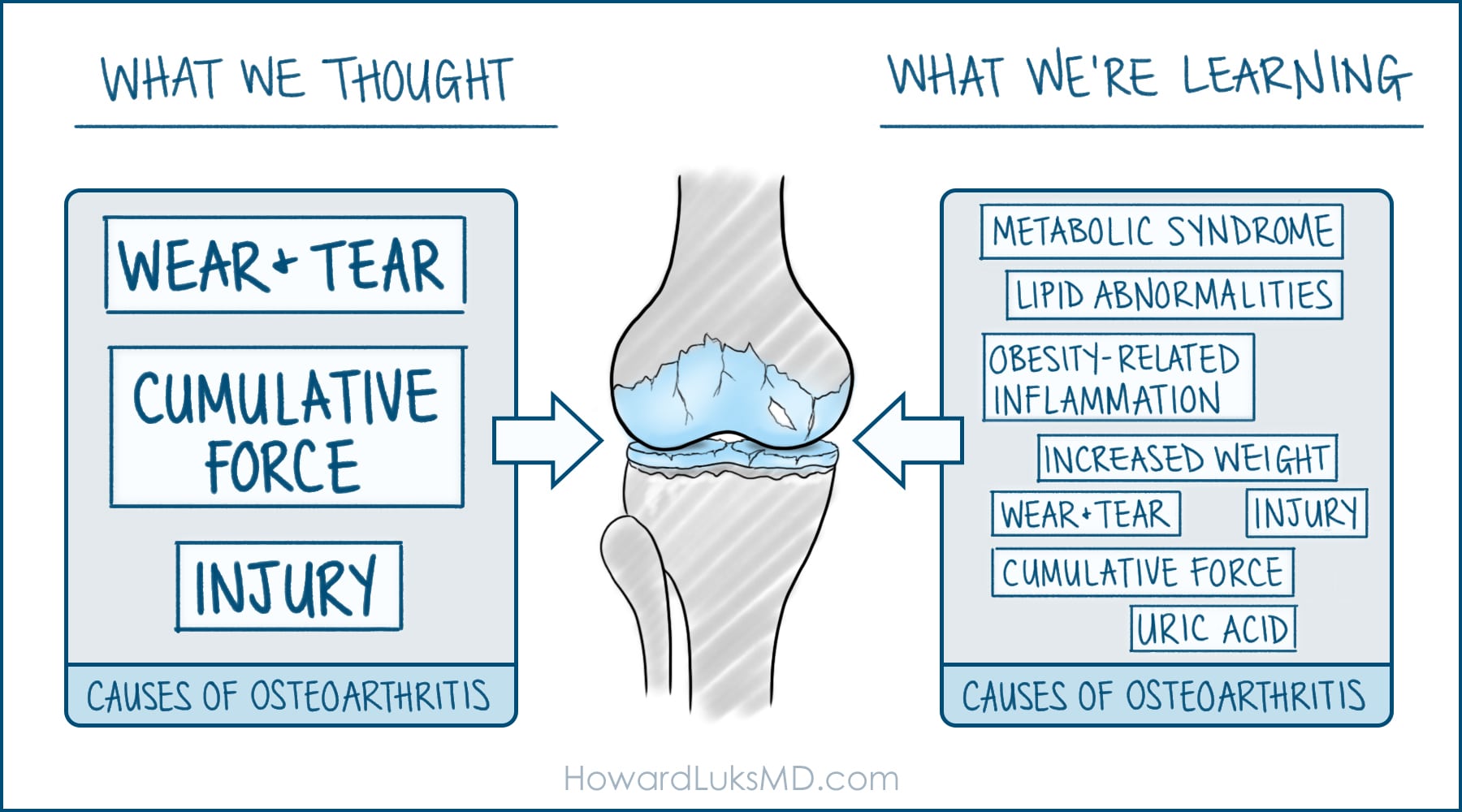
Source: HowardLuksMD.com
1. High glucose (blood sugar) levels can damage tendon collagen. Our tendons are filled with collagen (structural protein) fibers intended to withstand force. When force or stress is applied, these fibers unkink and slide to adapt. Over time, high glucose levels in the blood lead to the build-up of advanced glycation end-products (AGEs) on collagen fibers, causing cross-linking and stiffening. Animal studies show that these AGEs can inhibit tendon mechanics by limiting the fibers’ ability to unkink and slide. Recent research indicates that AGEs cause degenerative rotator cuff changes.
2. Insulin resistance leads to cartilage degradation: Research has shown that 82% of people with arthritis have an increase in blood insulin concentration and that insulin resistance—a hallmark of metabolic dysfunction and a signal that the cells can’t produce energy effectively—is mechanistically linked to cartilage issues. Key features of insulin resistance—including hyperglycemia, mitochondrial dysfunction, decreased AMPK activation, and chronic inflammation—are all associated with OA. Strategies that increase insulin sensitivity—like calorie restriction, physical activity, and some diabetes medications—are protective against OA.
3. Cholesterol deposits may weaken tendons. In a 2015 research review, study authors found a link between lipid levels and abnormal tendon structure or pain. The researchers theorize that high cholesterol may cause deposits to degrade the tendon matrix and drive inflammation. A recent cross-sectional study also found that cholesterol accumulation may reduce Achilles tendon function, potentially leading to injury. Dyslipidemia, a high level of lipids in the blood, is also associated with rotator cuff tears. And rotator cuff tendinopathy has a strong link to higher cardiovascular risk scores.
4. Worsening metabolic health causes inflammation. Chronic, unchecked inflammation reduces our body’s capacity to heal itself. Cholesterol and AGEs, as just discussed, can drive tendon and joint inflammation, but so can uric acid buildup in joints, which is also linked to insulin resistance. Research indicates that uric acid in synovial fluid, which surrounds joints, is linked to the cytokines (inflammatory proteins) interleukin-18 (IL-18) and interleukin-1 beta (IL-1β). In fact, uric acid-activated inflammasomes (inflammatory activators) produce these cytokines. Since IL-18 is linked with OA progression, researchers theorize that the immune system is involved in OA development. Oxidative stress, brought on by an imbalance in reactive oxygen species (ROS) production that can occur in metabolic dysfunction, also plays a role in OA development and progression by driving inflammation via cytokines.
5. Reduced blood flow to tissues, like joints, tendons, and muscles increases the likelihood of pain and damage. One of the most interesting lectures I’ve ever been to was by Jimmy Conway, MD, an orthopedic surgeon. The lecture was called “Hidden Connections: Tendon Health and Nutrition.” Dr. Conway stated that “rotator cuff tears are a heart attack of the shoulder.” Think about it: Metabolic health and insulin sensitivity determine how well our blood vessels function and how well they deliver oxygen to our tissues. If tissues (tendon, muscle, even cartilage) don’t get adequate blood flow, cellular dysfunction will arise, which leads to tissue dysfunction, like weak muscles, tendons, and ligaments. Good blood flow also delivers nutrients, hormones, signaling factors, and immune elements that can positively support tissue health. A well-oxygenated system is more resilient to damage—always. Metabolic health leads to better blood flow.
An expanded view of surgery for musculoskeletal issues
While surgery is sometimes needed to treat injuries or musculoskeletal conditions, it should rarely be something we jump to before conservative strategies. “Many of the issues that reveal themselves in our imaging studies are typical age-appropriate findings that do not require surgery,” notes Dr. Howard Luks, board-certified Orthopedic Surgeon.
And it’s important to remember that ALL surgery happens within the complex milieu and physiologic realities of a body, so anything we can do to optimize the body prior to surgery supports good surgical outcomes. For instance, optimizing blood sugar control around the time of surgery reduces postoperative infections and hospital stay length, and improves wound healing.
In some cases (but certainly not all), surgery may even do more harm than good. Other treatments may be just as effective while also being non-invasive. Plus, recovery from surgery can hinder our ability to move—at least for a while—potentially having detrimental effects on our metabolic health and well-being.
- Can surgery do more harm than good? Meniscus surgery is common for people with cartilage tears. The meniscus is a C-shaped cartilage structure that cushions the knee joint; each knee has two. In one study, researchers found that patients without OA who underwent meniscus surgery had a higher risk of developing OA and cartilage loss than those who did not undergo the procedure. The researchers found that all the 31 knees that had meniscus surgery during the previous year developed OA, but only 59% of the 280 knees with meniscal damage that did not undergo surgery developed OA. Of the surgery knees, cartilage loss occurred in nearly 81%, whereas it occurred in about 40% of those not operated on. As Dr Luks says, “the fastest way to generate osteoarthritis in a knee is to cut into it and take something out.”
- Is there a non-invasive option? A randomized-controlled trial in one of the top medical journals, NEJM, compared meniscus surgery outcomes to physical therapy outcomes, for example. The researchers randomly assigned 351 patients to either undergo surgery or physical therapy. All patients were 45 years or older and had a meniscus tear along with mild-to-moderate OA. At six-month follow-up, the researchers assessed the patients’ physical function scores. They found that those who underwent surgery had improved by an average of 21 points on the scale, whereas those in the physical therapy group had an average improvement of 18.5 points. Ultimately the results showed that improvement scores were rather similar between the two groups. About 30% of patients who underwent physical therapy then opted for surgery. At a 12-month follow-up post-op, improvement scores were still similar. The takeaway here is that surgery didn’t provide significantly enhanced function over physical therapy, and study authors concluded that “given that improvements in functional status and pain at 6 months did not differ significantly between patients assigned to arthroscopic partial meniscectomy and those assigned to physical therapy alone and that 70% of the patients in the physical-therapy group did not undergo surgery, these data provide considerable reassurance regarding an initial nonoperative strategy.”
- Plenty of people with structural issues in joints don’t have pain. Just because they have a torn piece of cartilage in their knee doesn’t mean a person will have pain or develop arthritis. Why? Because it is not just structural issues that contribute to symptoms, but also the biological issues of the joint like chronic inflammation, poor blood flow, or excessive pain-signaling activation. But these are all modifiable factors, so someone could have a structural issue but good joint biology, and therefore no pain. One study showed that 61% of people with incidental meniscus tears on MRI had no pain! Another study showed that 80% of people 60 years of age or older will show an incidental finding of disc degeneration on their spine MRI, even if they have no symptoms. Dr. Luks notes that when he was 50, he got an MRI of both knees just to see what was happening in there. He found that both knees had meniscus tears, and yet he is still a competitive runner, has no pain, and no arthritis.
Does movement make arthritis worse?
OA is not an inevitable part of aging or a consequence of exercise or movement. Instead, movement is one of the best things for our joints. In fact, Dr. Luks says exercise is one of the most effective treatments for early to moderate OA. The idea that exercising would further worsen the joint is a fallacy. He explains that exercise has anti-inflammatory benefits. One study showed that exercise increases interleukin-10 (IL-10), an anti-inflammatory cytokine that helps preserve cartilage. Plus, it decreases cartilage oligomeric matrix protein (COMP), a marker associated with OA. For the study, 31 women with knee OA participated. The exercise group of 16 women warmed up on a stationary bike and then performed leg-press sets on a machine. Their results were compared to a control group that did not exercise. Additionally, the impact of exercise can stimulate chondrocytes, the cells that actually make cartilage.
Another study looked at the potential benefits of 15 weeks of exercise in 19 women aged 60 or older who had sarcopenia (age-related loss of muscle mass) or sarcopenia with OA. After the program, the researchers found that levels of irisin (a protein) increased in the participants. Irisin has anti-inflammatory properties because it leads to a reduction in the release of pro-inflammatory cytokines. Not only that, irisin also helps protect against insulin resistance, Type 2 diabetes, and arteriosclerosis. And it encourages the browning of adipose tissue. Brown fat has more metabolic health benefits than white fat because it has a higher density of mitochondria. At the same time, participants’ levels of myostatin decreased. Myostatin (a protein) has been implicated in limiting muscle growth. Anti-inflammatory and cartilage-preserving IL-10 also increased, especially in the group with both OA and sarcopenia. Tumor necrosis factor alpha (TNF-a) decreased in this group, as well. In animal models, TNF-a has been implicated in the progression of OA and is a key pro-inflammatory molecule in the body associated with most chronic degenerative metabolic diseases like Type 2 diabetes and Alzheimer’s disease. Physical functioning scores improved in both groups of women in the study.
Muscle is thought to be the first tissue in the body to become insulin resistant, so moving the body and promoting muscle insulin sensitivity through exercise is a crucial way to manage whole-body glucose and insulin dynamics. The body is a unified system, so benefits that happen in the muscle can have wide-ranging effects on the whole body by promoting insulin sensitivity through tissue cross-talk.
Dietary changes and supplements may help the body heal
More research is needed on this topic, but since metabolic health and joint health are connected, improving metabolic health through diet may benefit your joints. Here’s what you can do:
Improve insulin sensitivity. Insulin resistance is associated with OA. But you can greatly improve insulin resistance by dialing in diet and lifestyle changes. Focus on nine elements of metabolically healthy meals that reduce glycemic variability and optimize the six factors that affect glucose aside from diet.
Eat more antioxidant-rich foods. Oxidative stress and resultant inflammation are linked to musculoskeletal and joint issues. Antioxidants are enzymes and molecules that can “scavenge” free radicals—unstable atoms with the potential to damage cells. They can also repair ROS-related damage. A diet rich in a rainbow of plant foods (fruits, vegetables, nuts, seeds, spices) will be high in antioxidants. You can learn more specifics about antioxidants in this post.
Consider supplements. One supplement with some evidence of benefit is turmeric (a spice). A research review investigating 10 studies found that supplementation with turmeric improved pain and function in participants with knee OA. In three studies, researchers were able to see a comparison between turmeric and non-steroidal anti-inflammatory (NSAID) medications. They found no difference in outcomes between the two. In all studies, no adverse events were reported regarding turmeric. NSAIDs, however, can increase bleeding and cardiovascular disease risk, and they may exacerbate asthma. In people with Type 2 diabetes, NSAIDs are also associated with an increase in hemoglobin A1c (a three-month average of blood sugar levels). Check out our Levels blog for some ideas on how to add more turmeric to your diet.
Other research has shown that specific doses of omega-3 fatty acids, vitamin C, Acetyl L carnitine, alpha lipoic acid, and B vitamins may all improve or augment OA and other joint pain. This makes sense in the context of these nutrients all being involved in key metabolic processes. See more about the Magic of Micronutrients for metabolic processes in my past newsletter.
Get a postbiotic boost. Urolithin A is a postbiotic produced by bacteria in your gut microbiome. It’s synthesized after eating ellagitannins and ellagic acid-rich foods. These include pomegranates, berries, and nuts, like walnuts and pecans—all rich in polyphenols, a type of antioxidant. In a recent study that analyzed chondrocytes (cartilage cells) from healthy knees, the researchers found that chondrocytes treated with urolithin A had improved mitochondrial function. Mitochondrial dysfunction is a hallmark of Type 2 diabetes and OA. Research indicates that not all gut microbiomes produce urolithin A effectively, so supplementation can be helpful. One randomized clinical trial of 66 people found that supplementation is safe.
We also have a mind-body connection to pain
Another thing I’ve learned through my joint injury journey and a history of back and neck pain (which I experienced during my surgical residency) is that there is also a strong relationship between mental health, metabolic health, and pain. Two books I’ve found helpful in understanding this more include Healing Back Pain: The Mind-Body Connection by John E. Sarno, MD, and The Body Keeps the Score: Brain, Mind, and Body in the Healing of Trauma by Bessel van der Kolk, MD. If you’re dealing with aches and pains, these may be worth a read. Mindset plays a role in controlling metabolism—as I discussed in another newsletter—and in many ways, impacts pain responses as well. Understanding and positively impacting our thought process and brain function can positively impact both our metabolic health and pain response.
Takeaway
In addition to the specific joint physiology discussed here, I want to reiterate the larger point: if we are to take a multimodal, holistic approach to healing seriously, we must be our own health advocates. We benefit from focusing on strategies that maximize the underlying physiology and biology of the body to support its healing and regenerative capabilities. This means optimizing metabolic health to support all aspects of health, including joint and musculoskeletal health. Even if your situation is one that requires pharmaceutical or surgical intervention, a body that is healthier and more metabolically functional because of optimized daily habits around unprocessed food, exercise, sleep, stress, light, and toxin exposure will do better.
There is also something to be said for learning to lean into the discomfort of uncertainty. Having an option right in front of you—a pill or surgery—can feel like a silver bullet (and sometimes they can be!), while a non-invasive approach with an unknown endpoint for healing can feel uncertain. Certainly, in the couple of months after the injury, when I was limping and my knee was still swollen and making clicking and grinding sensations with every step, it was scary and disconcerting, and I shed a few tears of fear not knowing when I’d be back to full steam. But it’s worth considering whether our desire to reach for quick fixes is in part to minimize the discomfort of waiting and whether doing so can ultimately undermine the body’s ability to activate innate healing paths that benefit the whole system. Part of healing—with or without surgery—is believing that your body can heal.
In many cases, surgery may be the necessary treatment and it may need to happen immediately. But if I can find ways to encourage my body to heal itself, I will always opt for that method first or in conjunction with more aggressive therapies.








