Calories In/Calories Out Is A Flawed Model
The process of gaining weight isn’t as simple as one might think. The model of “calories in/calories out” that many of us are familiar with is flawed and doesn’t consider the complex hormonal and biochemical pathways involved in energy balance, weight gain, and weight loss in the body.
The flaws of the calorie deficit model of weight loss can be pictured by imagining the human body as a car: The engine is our cells, the fuel tank is our fat stores, and the fuel within the tank is provided by the food we eat. As we drive the car, the engine (cells) burns fuel (fat) to keep the vehicle moving. By consuming food, we “refill” the tank as we go, preventing it from running dry. In this analogy, if we want to deplete our fuel tank (fat stores), we can put less fuel in the tank (eat fewer calories) or drive the car faster (exercise more).
“Different types of foods eaten at different times of the day and in different combinations can lead to unique hormonal responses in the body.”
That’s all straightforward and makes sense at first blush, but is it accurate? Is a car with just three components a reasonable way to represent the complexity of the human body? Moreover, is a car with three components even a reasonable way to represent a real car’s complexity? The answer is no, and here’s why: A real engine doesn’t just require fuel to run – it requires a precise blend of fuel and oxygen, along with a perfectly timed spark, controlled temperature and humidity, lubrication, and more. Furthermore, every engine model might be constructed slightly differently. Tip the balance on any of these mitigating factors, and the fuel efficiency will change dramatically if it manages to run at all.
The original thought experiment now seems a lot more complex. What happens if we drive this more realistic car, except this time debris has clogged the air filter, preventing oxygen from entering and making conditions impossible for the fuel to burn efficiently? What if the engine oil is depleted, and there is excessive friction and wear on the engine parts? This is a more accurate representation of the human body and the hormonal mechanisms of weight and energy balance.
Amazingly, different types of foods eaten at different times of the day and in different combinations can lead to unique hormonal responses in the body, and it is these hormones–in particular, insulin–that determine what happens to the food molecules once broken down by the digestive system, and how efficiently they are processed. Moreover, different people can have highly variable glucose responses to the same food, regardless of calorie content.

How a CGM can help you find your optimal diet and lower blood sugar
There’s no "ideal" diet for everyone, but a continuous glucose monitor gives you real-time feedback on the foods that work best for your metabolic health.
Read the ArticleThe Role Of Insulin
Insulin is released in response to glucose in the blood (see Figure 1) and is the primary hormone involved in fat storage and weight gain. Though we often associate insulin with diabetes, it is a big contributor to weight gain for everyone–with diabetes or not–and has to be tamed to maintain a healthy weight successfully.
Insulin is our body’s primary anabolic hormone, meaning it promotes “building” in the body (e.g., the storage of fat) rather than breaking things down. Insulin tells our cells to take up glucose from the blood for use or storage if there’s excess. Because weight loss generally requires us to burn through fat stores, we need to control our insulin to signal the body that it should burn fat rather than store more.
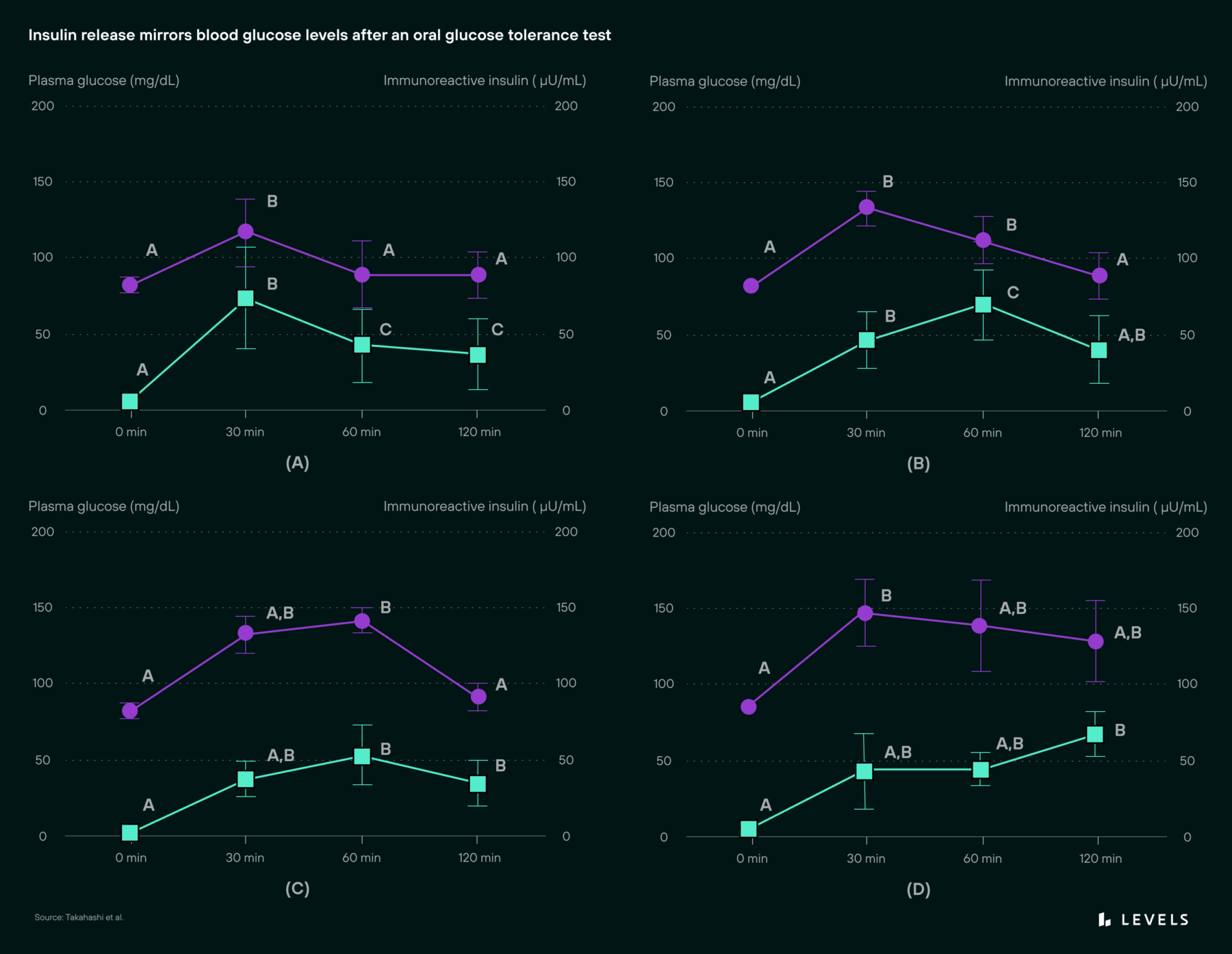
Figure 1: Insulin release mirrors blood glucose levels after an oral glucose tolerance test. Source: Takahashi et al.
When more glucose is in the bloodstream than the body needs to meet energy demands, increased insulin levels signal to the liver and muscles to store glucose in chains of glycogen. Once the liver and muscles are filled to the brim with glycogen, the excess glucose is turned into fat (triglycerides) and sent out in the blood to be stored in the fat cells around the body. You can think of the liver and muscle as short-term, limited storage space for energy in the form of glycogen and fat cells as long-term, essentially unlimited storage space for energy in the form of triglycerides.
When we’re not eating– between meals, sleeping, or fasting–the lack of dietary glucose causes insulin levels to fall. This signals our body to burn stored energy, starting with glycogen. Once glycogen stores run out, we start burning fat (see Figure 2). The goal of weight loss is to burn excess stored fat by mobilizing it and bringing it to parts of the body that can use it for fuel. It’s important to note that insulin’s anabolic (storage) signal prevents the body from tapping into fat for energy as long as insulin levels are high.
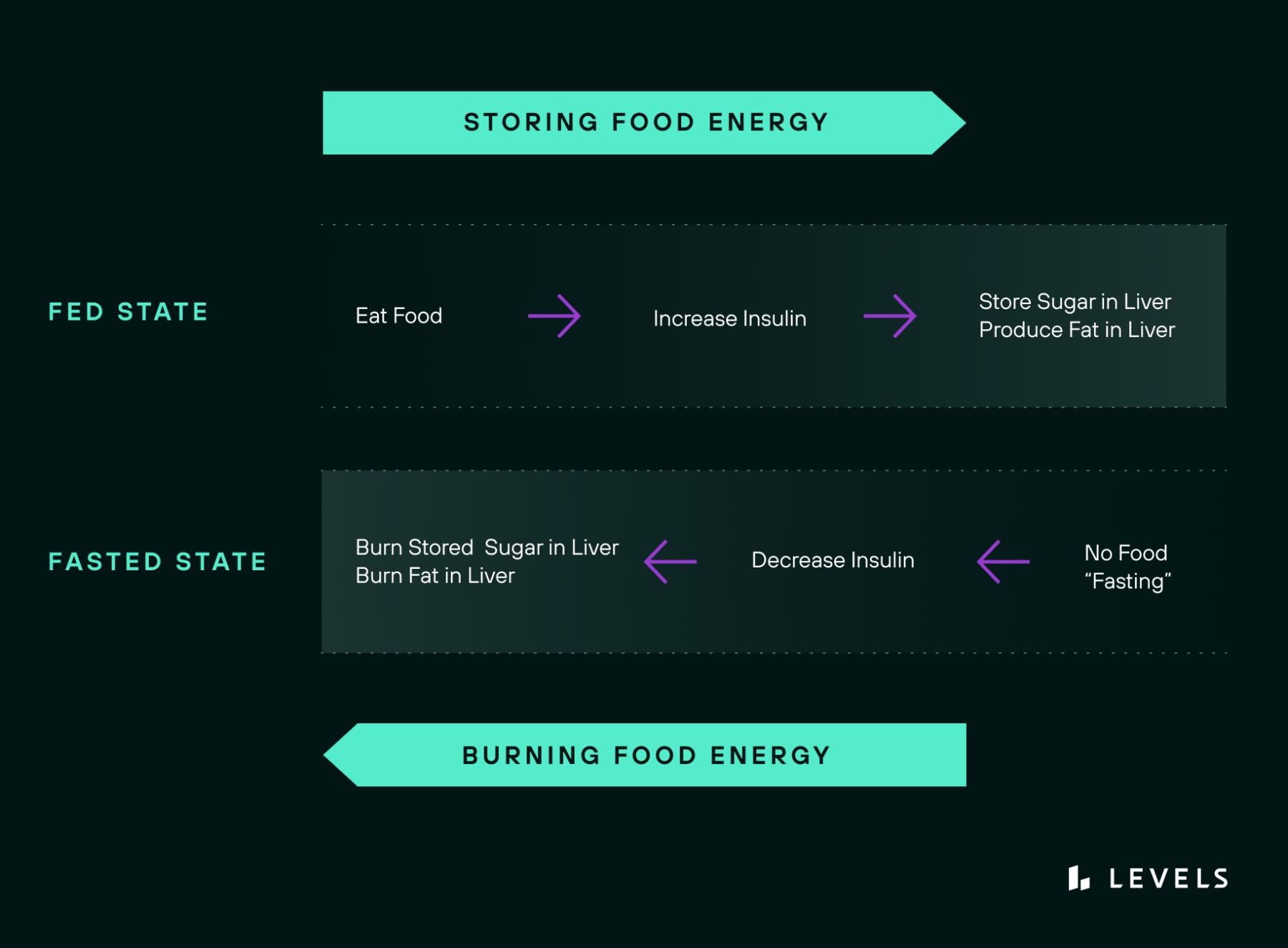
Figure 2: Eating food that raises glucose leads to an increase in insulin, which tells the body to store sugar in the liver and produce fat from any excess. While fasting, our insulin levels decrease and this facilitates burning fat and stored glucose. Figure credit: Jason Fung, MD.
This part of the story is simple: we need to lower our insulin levels to burn stored fat. But what if our insulin levels never really fall enough to signal that we should be burning through our stored fat? This scenario is similar to the car with the clogged air filter – we can push the accelerator all we want. Still, unless the oxygen (insulin) level is optimal, the engine (cells) can’t burn fuel (fat) efficiently. In the body, high insulin impairs triglyceride breakdown in fat cells and inhibits fatty acids from entering into fatty acid oxidation in the mitochondria.
Learn More:
The Importance Of Understanding And Controlling Our Glucose Levels
At Levels, we believe knowing exactly how your body responds to different foods is the key to success. Fortunately, glucose levels tend to mirror insulin responses. If we know how much a food raises our blood glucose, we have a more robust understanding of our body’s predicted exposure to insulin and, therefore gain a sense of whether our body is likely to be in a fat-storage or fat-burning mode.
Reading this, you might think we should just eat very few carbohydrates and stick to high-fat foods to decrease insulin and lose fat and weight. This may be part of the puzzle, but it’s not the only piece. It’s important to remember that many complex carbohydrates and healthy protein sources can help our bodies function optimally and may not generate significant glucose and insulin spikes.
The “glycemic index” — a ranking of foods based on how they affect blood glucose — is a standardized tool to help understand this process, but it can also be a blunt instrument. A few problems have become increasingly clear regarding standardized glycemic indices:
- Interpersonal variability: Different people who eat the same meal might have highly variable glucose responses due in part to the unique and complex biologic environment of their bodies.
- Intrapersonal variability: Different meals with the same amount of carbohydrates can generate highly variable glucose levels in a single person. Carbohydrate content alone is often a poor predictor of personal glycemic response.
- Lifestyle: Glucose levels might change based on factors such as stress and sleep quantity. For example, restricting sleep to 4 hours per night for 6 days has led to higher glucose response to specific foods and a 40% lower rate of glucose clearance from the blood.
- Timing variability: The time of day we eat can profoundly impact glucose levels in the blood, with a high-glycemic and high-calorie meal eaten in the evening causing significantly greater glucose and insulin response compared to the same meal consumed in the morning.
- Food combinations: The meal’s composition and the order of foods consumed can cause glucose to change differently than if those foods were eaten in isolation (See Figure 3).
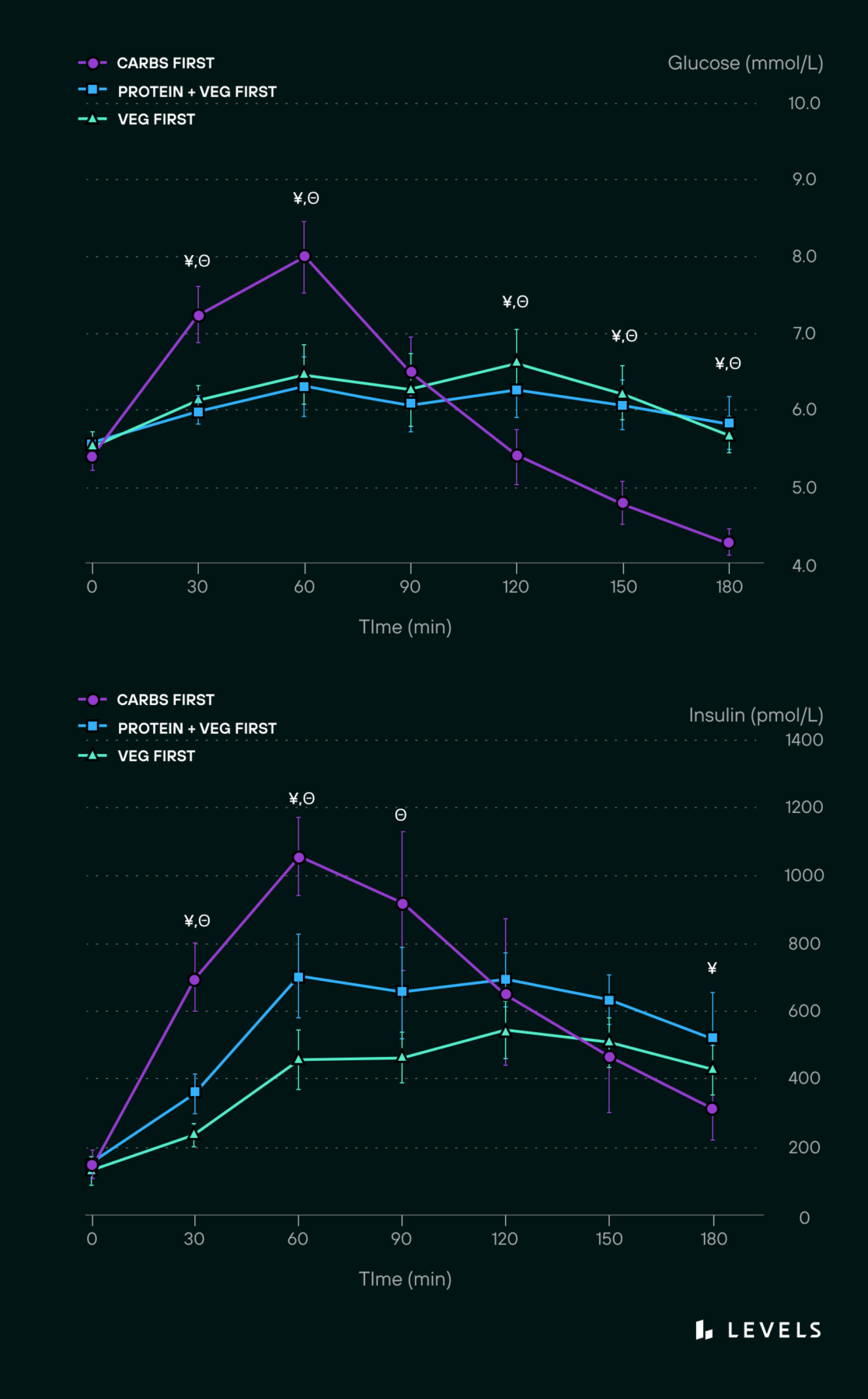
Considering all these variables, how could someone possibly know how their day-to-day diet affects their glucose levels if they don’t have access to their glucose data? The answer is simple: They can’t.
Why 85% Of Diets Fail
Unfortunately, long-term success rates with weight loss are dismal: despite the alarming statistic that 70% of Americans are overweight or obese, and nearly 50% of Americans have tried to lose weight in the past 12 months, only 15-20% of people who successfully lose weight can keep it off. As stated by Ochner et al., “this almost ubiquitous weight regain is witnessed in virtually every clinical weight-loss trial, including those specifically aimed at improving weight loss maintenance.”
Why do conventional attempts at weight loss fail? Three things: metabolism, hormones, and the brain. The reality is that our bodies have evolved to help us survive and hold on to energy in the face of starvation through various complex mechanisms.
Simply eating less (i.e., calorie deprivation) is a common approach to weight loss but thwarts our efforts by reducing our resting metabolic rate. When the body senses an environment of food scarcity, it uses energy more efficiently and reduces its use of stored energy. This translates to fewer calories expended per day.
The types of foods we eat after losing weight also seem to affect our ability to maintain weight loss. A study showed that people who adhere to low carbohydrate eating plans after weight loss burn ~200 kilocalories more daily than those on higher carbohydrate diets. For those with the highest insulin secretion at baseline who subsequently adhere to a low carbohydrate diet, that difference widens to 400 kilocalories per day.
The brain is also affected by diet in ways that promote weight regain. When we are calorie-deprived, we see increased brain activity that leads to increased attention, reward, and motivation related to food. In other words, calorie deprivation makes us hyper-focused on obtaining food.
Hormones, including those beyond insulin, play a significant role as well. Leptin is one of our appetite-inhibiting/satiety hormones and is secreted by fat cells in response to eating. Its levels also increase as fat mass increases. It is thought that leptin signals to the brain to inhibit food intake to prevent overconsumption of dietary energy and suppress insulin production to discourage further fat storage in favor of fat burning.
Paradoxically, obese patients with higher levels of leptin (due to higher levels of fat mass) may suffer from “leptin resistance,” thought to be due to reduced transport of leptin across the blood-brain barrier. High insulin levels are also thought to lead to leptin resistance, preventing appetite inhibition and leading to a cycle of increased weight gain.
How Glucose Tracking Can Inform Our Weight Loss Efforts
It all starts with glucose levels – if they’re elevated, insulin is generally released. Increased insulin levels lead to fat storage and weight gain. Over time, if glucose is consistently elevated due to diet and insulin production is constantly active, our cells can become “numb” to insulin, which is called insulin resistance. This means that we need more and more circulating insulin to get glucose into cells, leading to higher baseline levels of insulin. This process directly counters weight loss efforts.
In obese people, high insulin levels also lead to impaired leptin signaling and “leptin resistance,” making it harder for us to feel full and more likely to continue consuming calories rather than burn stored fat for energy. On the other hand, decreased glucose levels lead to lower insulin levels, leading to weight loss and decreased fat mass.
Real-time glucose measurements give us the power to understand how the foods we eat affect glucose levels in our blood and, by rough proxy, our insulin levels. Unlike traditional dietary strategies like calorie counting — which have been shown repeatedly to be ineffective for sustained weight loss — glucose monitoring provides insight into the underlying physiological processes that lead to fat storage.
Closing Thoughts: Why Should We Even Care About Losing Weight? Spoiler: It’s Not Just About Looks.
Being overweight is a risk factor for many problems: cardiovascular disease, cancer, diabetes, and premature mortality. It can also exacerbate hypertension, arthritis, gallstones, high cholesterol, low back pain, bronchitis, and musculoskeletal problems.
Excess fat also acts as a source of many signaling molecules in the body and is even thought to be an “endocrine organ” in its own right. Fat can secrete hormones (like those associated with appetite, i.e., leptin) and pro-inflammatory chemicals called adipocytokines, which are associated with insulin resistance. Visceral fat — the type that surrounds organs and is most dangerous in terms of risk for chronic disease — is also known to harbor many immune cells called macrophages, leading to further production of pro-inflammatory chemicals.
Given the relationship between glucose, insulin, and excess fat, real-time monitoring can provide a valuable tool to help people feel more empowered in their weight loss journey. Personal data helps people know how their diet affects them and can inspire behaviors promoting optimal weight and long-term health and wellness.
 Want to learn more about your metabolic health?
Want to learn more about your metabolic health?
Levels, the health tech company behind this blog, helps people improve their metabolic health by showing how food and lifestyle impact your blood sugar, using continuous glucose monitoring (CGM), along with an app that offers personalized guidance and helps you build healthy habits. Click here to learn more about Levels.


 Want to learn more about your metabolic health?
Want to learn more about your metabolic health?


