How many times have you heard the word ‘antioxidant’? Maybe you read it on the back of an orange juice carton or saw it on a supplement bottle. That wouldn’t be surprising: The term has been called a “nutrition buzzword” since at least 1994. But aside from having a vague idea that antioxidants are healthy, most people don’t know what antioxidants do.
“Understanding how antioxidants can protect us from oxidative stress, and the diseases linked to it, comes down to comprehending what’s happening on a microscopic level inside your cells.”
Antioxidants are naturally occurring or human-made substances that protect against oxidative stress—a state of imbalance in your cells linked to diseases including cancer and diabetes.
Decades of research suggest that antioxidants form the basis of a defense system within the body that can neutralize specific rogue molecules (more on that later). Understanding how antioxidants can protect us from oxidative stress, and the diseases linked to it, comes down to comprehending what’s happening on a microscopic level inside your cells.
What is oxidative stress?
When scientists describe oxidative stress, they often use a seesaw as an analogy. On one end of the seesaw are antioxidants. On the other are molecules called reactive oxygen species, or ROS for short.
ROS are formed when a molecule’s electrons go rogue. Electrons are tiny, negatively charged subatomic particles found in all atoms that usually come in pairs. In their paired state, they are relatively stable and nonreactive, which means they don’t interact with other molecules in the body and thus don’t trigger any unwanted processes.
Occasionally, one electron breaks free of its pair, or, most often, electrons leak out from parts of the cell that harbor many of them. For example, mitochondria, the cell’s energy-producing powerhouses, are rife with electrons.
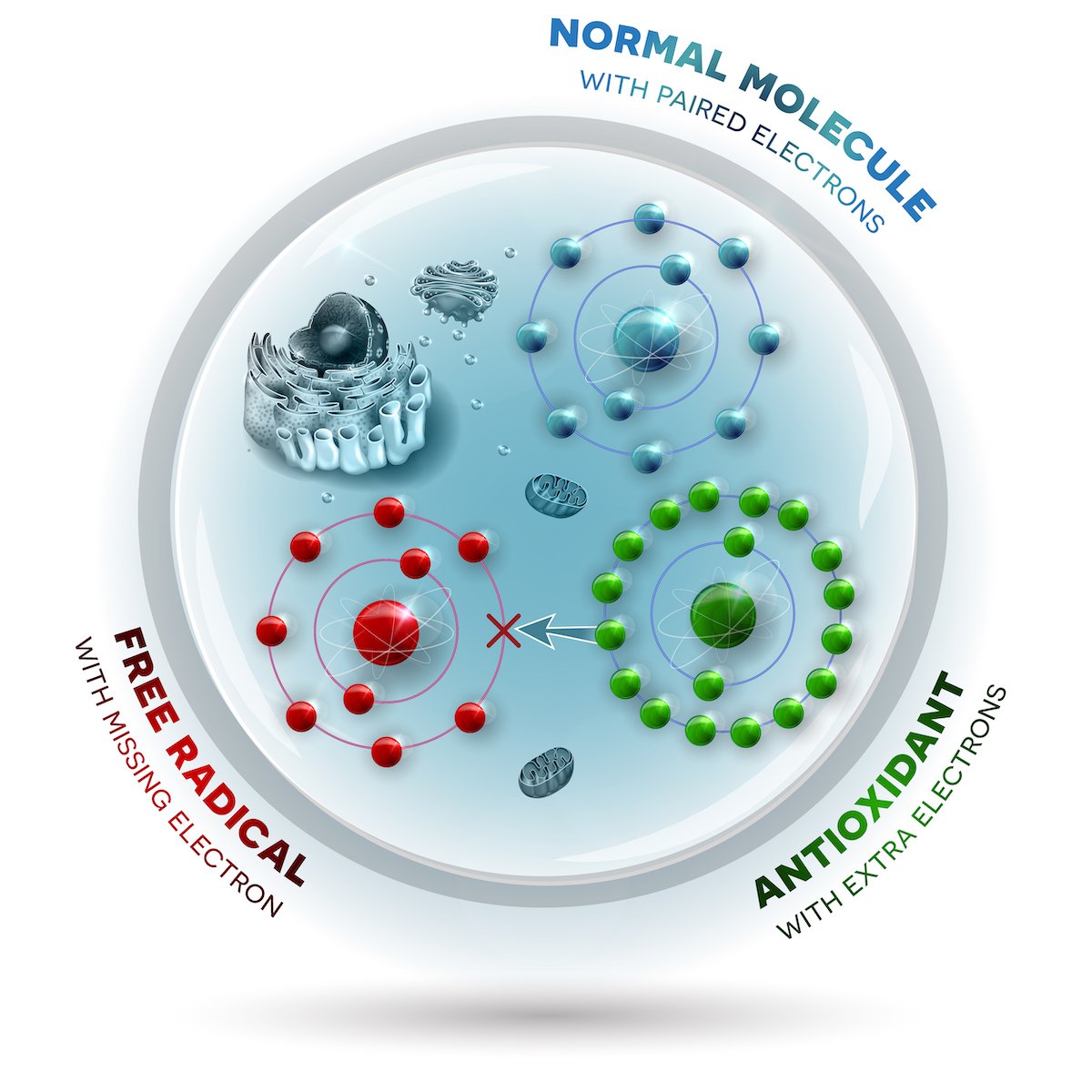
These unpaired electrons can occasionally bind up with other molecules, like nitrogen or oxygen. Armed with that extra, rogue electron, these molecules become reactive—that is, capable of interacting with other molecules and sparking downstream effects. When nitrogen picks up an extra electron, it’s called a reactive nitrogen species (RNS); when oxygen does this, it’s called a reactive oxygen species (ROS).
In normal circumstances, ROS are created when cells manufacture energy within the mitochondria. Antioxidants, both produced by the body and obtained through certain foods, can manage ROS in several ways, from scavenging up ROS and rendering them harmless to breaking chemical chain reactions that produce extra ROS. In short, they can work at several different levels to stop ROS from proliferating and causing damage. To go back to the seesaw analogy: ROS and antioxidants typically exist in a state of balance.
ROS become dangerous when they either accumulate or become entirely depleted. High concentrations of ROS can damage proteins, fats, or nucleic acids (like those found in DNA) and lead to cell death. Especially low concentrations of ROS may also derail healthy cell signaling pathways. In these cases, the seesaw tilts too far in either direction. This unbalanced state is called oxidative stress.
The imbalance between ROS and antioxidants can create conditions inside the body that lead to disease. For example, excess ROS allows cancer cells to thrive in the body.
In other cases, when ROS overwhelms antioxidant systems, it can lead to heart disease. ROS can bind with LDL (sometimes called “bad” cholesterol), the resulting “oxidized LDL” can kill certain cells within arteries or attract different types of cells into the vasculature, leading to the formation of a tough, more advanced form of plaque, which causes the arteries to narrow.
Studies have also linked excess levels of ROS to complications related to diabetes or the development of neurodegenerative diseases.
While the connection between ROS and disease is well established, the role of ROS in the body presents a bit of a puzzle. Why have ROS survived thousands of years of evolution if they’re such a threat? It’s because ROS also play a role in healthy cell signaling.
ROS are known as “secondary messengers” within the body—molecules that relay signals within cells (in contrast, “first messengers” relay signals between cells). When in balance with antioxidants, ROS set normal cell pathways in motion and even use their destructive abilities to target invaders. For example, one 2010 review paper points out that ROS may help the body destroy harmful microbes. At “tolerable levels,” ROS may even work as anti-inflammatory agents, as one 2020 review paper in Autoimmune Disease explains.
So, evolutionarily speaking, ROS are a form of compromise. On the one hand, they’re potent signaling molecules. But on the other hand, if they’re too abundant, they set off a state of oxidative stress.
This means that combating the ill effects of oxidative stress, from inflammation to metabolic complications, is not as simple as boosting antioxidants to soak up ROS. Instead, it’s about fine-tuning a delicate cellular balance to keep them in check.
What causes oxidative stress?
Some ROS production is natural in a healthy functioning cell. When your body combines oxygen and food sources to make energy, it passes electrons along a long chain of proteins embedded within the membrane of mitochondria, the energy-making portion of a cell. Most of the time, electrons are used up in this process, known as the electron transport chain, to create energy in the form of a molecule called ATP, together with water as a byproduct.
But occasionally, electrons slip out from that chain. When this happens, they can react with nearby oxygen and form ROS.
Typically, that’s not anything to worry about, provided that the body’s antioxidant defense systems are strong enough to neutralize ROS or there’s not an excessive amount of ROS floating around. But if either antioxidant systems are depleted or ROS accumulates, damage can happen.
There are several ways that ROS can be introduced into cells, tipping the balance beyond what the body can handle on its own. Here are a few ways ROS can accumulate:
Overeating
ROS production is triggered when we consume most major macronutrients (carbs, fats, proteins). That may be one reason why caloric restriction has been linked to lower ROS generation in animal studies. However, oxidative stress is caused mainly by extreme eating patterns.
Not eating enough and malnutrition have been shown to increase ROS levels beyond the realm of physiological balance. Overeating can have the same result. Overnutrition (overeating that leads to weight gain) triggers processes in the cell that lead to ROS production.
For example, when the body has too many nutrients to process, the mitochondria’s electron transport chain ends up with extra electrons, which can escape to form ROS. And when physical activity is low, less of those nutrients are used to make energy, compounding the electron glut. When we overeat, the electron transport chain is overburdened and, therefore, leakier. And when those electrons escape, more ROS form.
Studies in humans have linked overeating and ROS production. For example, in a week-long study on 10 healthy men in Korea, five of the men ate normally, while the other five ate about 3,500 calories per day in high-fat and high-carbohydrate meals. The high-calorie group showed an uptick in ROS production and antioxidant production, but overall, they generated a more significant amount of ROS than antioxidants. In other words, their bodies couldn’t compensate for the extra ROS generated by that week of extreme overeating.
Other papers consistently link excess nutrition to oxidative stress. For example, one review paper found an association between postprandial glucose and lipid levels to oxidative stress across nine studies investigating this link. The biomarker most closely linked to oxidative stress was the level of post-meal triglycerides in the blood (fats that the body uses to store extra calories).
The big picture is: When the body is in a post-meal state, unused nutrients can cause oxidative stress.
Poor Diet
Oxidative stress isn’t just a factor of how much you eat. What you eat influences the state of your cells as well.
Animal studies have linked diets high in sugar and fat to oxidative stress. In one study, mice fed high-glucose diets for four weeks had decreased levels of key antioxidants, and their genes related to greater ROS production were switched on. With their antioxidant defense systems weakened and ROS-producing fires stoked, these mice entered a state of oxidative stress (they also developed diabetes, which the authors propose could have been set in motion by oxidative stress).
Some review papers examining the impact of specific diets on oxidative stress have found that certain eating patterns have greater impacts on antioxidant-ROS balance than others. For example, the “Western Diet” or “fast food diet” consisting mainly of red and processed meat, dense caloric food, refined carbohydrates, and added sugar is linked to increased oxidative stress biomarkers. These biomarkers include DNA, lipids, proteins, and carbohydrates that have interacted with ROS, and antioxidants that have changed due to increased ROS. Other eating patterns seem to be less harmful. Compared to the Western diet, the Mediterranean diet (consisting mainly of olive oil, vegetables, white meat, fish, fresh nuts, and seeds) was linked to fewer signs of oxidative stress and a greater presence of antioxidant defenders. Similarly, people who follow vegetarian diets have been shown to have lower ROS and other oxidative stress biomarkers.
The way some foods are prepared can also impair the body’s antioxidant defense systems, setting the stage for oxidative stress. For example, animal studies suggest that re-heated vegetable oils (which might be used for deep frying) decrease antioxidant levels. Oils with higher levels of unsaturated fatty acids (polyunsaturated fats like linolenic acid and linoleic acid) oxidize more quickly because of their chemical structure, which contains more double bonds than monounsaturated or saturated fatty acids. Some examples include vegetable oils like soybean, sunflower, and safflower oil. They also oxidize more quickly when heated. The health risks of consuming oxidized oils, however, are still up for debate. A pair of 2007 reviews in the journal Molecular Nutrition and Food Research presented both sides of the argument. One cautioned that regularly consuming oxidized oils posed a “chronic threat” to human health because it led to widespread inflammation, while the other argued that there was little high-quality evidence that the compounds in these oils appear in high enough concentrations to be toxic and that the body was well equipped to protect itself from any ill effects through antioxidants and detoxifying enzymes.
The effects of protein, fat, and sugar
Studies have shown that macronutrients—protein, fat, and sugar— can contribute to oxidative stress in different ways.
For example, in one highly unappetizing clinical trial done in 2002, 15 men and women fasted overnight. They then drank half a cup of either straight cream (high in fat) or a mixture containing casein (a protein found in milk). In both cases, the ROS levels of participants rose after eating. But after three hours, ROS levels in those who drank the protein shakes returned to normal, while the cream group’s ROS levels remained elevated.
Other research has shown that glucose consumption can lead to ROS production, too, through more than one mechanism. High blood sugar can lead to the production of triglycerides, high levels of which are linked to oxidative stress. But it can also set other ROS-related pathways in motion. [More on these mechanisms below.]
One crucial pathway appears to involve the production of uric acid—a waste product found in blood. When we consume fructose (a type of sugar), the liver uses a lot of energy (in the form of ATP) to break it down. That process sets off a chain of reactions that prompts the body to produce even more uric acid, which has consistently been linked to the development of oxidative stress in cell and animal studies.
Alcohol
There are a few ways alcohol can prompt the body to create more ROS. One is that ROS are produced when alcohol gets broken down by the body. As alcohol is metabolized, the body forms byproducts that boost the activity of the electron transport chain, resulting in the overproduction of ROS.
Meanwhile, other studies have shown that even alcohol can decrease the levels of certain antioxidants and increase biomarkers of oxidative stress. For instance, a 2004 European Journal of Clinical Nutrition study on 53 post-menopausal women who consumed 30 grams of alcohol per day for eight weeks (about two drinks) found that levels of α-tocopherol (an antioxidant) declined while markers of oxidative stress increased.
Inflammation
Inflammation and oxidative stress tend to go hand in hand. When the immune system senses trouble—like a harmful pathogen—it releases an army of immune cells in response. Those cells release enzymes, chemicals, and ROS, which can help destroy invaders, in part by causing inflammation. But if the body is constantly in a state of inflammation, this cycle of ROS production can become sustained, leading to oxidative stress.
The presence of extra ROS can have a snowball effect. It can actually trigger more inflammation because those ROS cause the cell to send out more inflammatory cytokines (chemical messengers than recruit more immune cells), perpetuating inflammation itself. In this regard, oxidative stress is both a precursor to this type of persistent inflammation and a consequence of it.
Stress
Psychological stress is increasingly recognized as a factor leading to oxidative stress.
When the body is under stress, perhaps due to an invading pathogen, immune system messengers, called cytokines, begin to flow. Psychological stress can also trigger the release of cytokines. As one 2018 review paper argues, those cytokines are major “promoters” of oxidative stress because they trigger ROS release.
Studies of real-life stressors have linked psychological stress to oxidative stress. One 2004 study analyzed the blood of 15 medical students before and after big exams to find oxidative stress biomarkers. In the days leading up to the exams, the students had lower antioxidant levels and higher levels of DNA and lipid damage. This suggested that oxidative stress had already swept through their cells during this stressful time.
Job stress has also been shown to contribute to oxidative stress. A study conducted in Japan, for example, showed a correlation between 8-hydroxydeoxyguanosine (8-OH-dG), a marker of oxidative stress, and female workers’ perceived workload, psychological stress, and impossibility of alleviating that stress. A similar study in Spain found a correlation between malondialdehyde, another oxidative stress biomarker, and high levels of work-related stress.
Cigarette smoke
Cigarette smoke is both a notorious ROS generator as well as a source. High levels of ROS are found in cigarette tar, and thousands of chemicals present in cigarette smoke can trigger the body to produce extra ROS on its own, for example, by breaking down mitochondria. Inhaling cigarette smoke is associated with increased oxidative stress and also overwhelms the body’s antioxidant defense system, causing antioxidant levels to decline. Over time, those antioxidant levels don’t seem to bounce back. Studies have shown that smokers have lower levels of certain antioxidants than nonsmokers.
UV light
UV light from the sun doesn’t contain ROS, but specific kinds of UV can trigger the body to produce ROS on its own. About 95% of the UV light you encounter is UVA light, which has a longer wavelength but less energy than UVB and UVC. It also generates more ROS.
UVA light can penetrate the skin and reach specific molecules that absorb light, like melanin or DNA itself. These molecules absorb light as energy, sometimes putting them into an “excited” or higher-energy state. They, in turn, can excite nearby molecules, including oxygen, generating ROS.
Pollution
Air pollution can also mess with the balance between ROS and antioxidants, leading to oxidative stress. That’s especially true of one type of air pollution called PM 2.5—fine particles that pose the greatest health risks because they can penetrate deep into the body. These particles can themselves contain ROS but also can trigger the generation of ROS inside the body by activating macrophages, mitochondria, and ROS-producing enzymes.
Several studies have shown that the components of air pollution, which include metals or other compounds, generate ROS within cells as they penetrate the body. According to one 2018 review, particulate pollutants can be considered “potent oxidants” and may usher cells towards early death.
Oxidative Stress and Metabolic Health
Research increasingly shows that high blood glucose, or hyperglycemia, sets off a chain of reactions leading to oxidative stress. Some research suggests that hyperglycemia’s ability to trigger ROS production and inflammation is a driving factor behind some of the complications linked with diabetes, like vision loss, nerve damage, kidney problems, or accelerated coronary artery disease.
How high glucose leads to oxidative stress
Studies have shown that sustained hyperglycemia can cause a cascade of events that lead to more ROS within cells: For example, high glucose levels can lead to increased production of the molecule diacylglycerol, which activates an enzyme called protein kinase C. This activates the enzyme NADPH-oxidase, which in turn can convert oxygen into ROS. This process has been called the “dangerous metabolic route in diabetes.”
High glucose can lead to oxidative stress through several other pathways. It has been shown to impair the activity of some antioxidants, lead mitochondria to make too much ROS, and activate the polyol pathway, which causes the enzyme NADH oxidase to produce ROS. In essence, all these pathways do the same thing: They either cause the cell’s machinery to create more ROS or deplete antioxidants.
Effects of chronic vs. acute hyperglycemia
Chronic hyperglycemia triggers harmful ROS production, but acute glucose spikes do as well. Research on 21 people with Type 2 diabetes published in JAMA found that blood glucose spikes after eating correlated with signs of ROS production and had a “more specific triggering effect on oxidative stress” than chronic hyperglycemia. Fluctuations in blood glucose were measured by calculating the mean amplitude of glycemic excursions (MAGE) over 24 hours, which was on average 75 mg/dL among participants with diabetes. To explain their findings, the authors situated them within a growing body of research linking diabetic complications specifically with rapid swings in glucose (as opposed to chronic hyperglycemia), which can partially be explained by the activation of ROS generation. One study they pointed to established that post-meal hyperglycemia induces overproduction of superoxide.
Studies suggest that managing glucose swings can lead to fewer signs of oxidative stress. In one trial done in 2012, 90 people with diabetes were given drugs to lower blood glucose levels. After 12 weeks, the participants had smaller daily glucose swings as well as fewer signs of oxidative stress than they did when the study began.
Oxidative stress and insulin sensitivity
Finally, oxidative stress may be one driver of insulin insensitivity, a significant factor affecting metabolic health. An existing theory suggests that oxidative stress damages some proteins involved in the insulin response, leading to insensitivity. The research, however, is mixed: As Ben Bikman points out in his book, Why We Get Sick, people with insulin resistance show more oxidative stress biomarkers than those who don’t, but treating insulin resistance with antioxidants doesn’t always lead to improvements.
In one small study published in Science Translational Medicine, six healthy men were told to eat 6,000 calories per day for a week and do nothing but stay in bed. Overall, the men gained about 3.5 kilograms per week, and their cells also showed higher oxidative stress markers.
The participants’ blood samples showed changes to a protein called GLUT4, an insulin-activated transporter that brings glucose into cells. The changes made the transporter dysfunctional, which the authors interpreted as a sign of insulin resistance.
This study had some significant limitations: it was small, had no control group, and didn’t determine which came first, insulin resistance or oxidative stress. Other studies, however, have suggested that oxidative stress triggers insulin resistance down the line, which led the authors of this study to argue that oxidative stress was probably the “initial event” predating the development of metabolic issues.
What can you do about ROS?
Accumulating ROS and oxidative stress aren’t inevitable. There are two primary strategies for controlling ROS: upping the level of antioxidants in your system (by turning on the body’s antioxidant-making pathways or getting them through food) and avoiding triggers for more ROS production, like smoking or excessive air pollution.
Keep in mind there’s no way to totally sidestep the process of producing ROS, and it wouldn’t be a great idea to do so because ROS at balanced levels are part of a natural and healthy functioning cell.
Stoking the body’s “master regulator” of antioxidants
Antioxidants are enzymes and molecules with the power to “scavenge”—seek out and neutralize other molecules that may cause harmful reactions. They can also bind to certain metals (that promote ROS formation) or act as cellular repair crews that clean up ROS-related damage.
Some antioxidants are formed through certain pathways within cells. Their so-called “master regulator” is the Nrf2 pathway, which is a chain of events that tells the body to switch “on” specific protective genes. Those genes, in turn, are translated into antioxidants that can detoxify and eliminate ROS. Eating certain foods can activate the Nrf2 pathway.
Some scientists have proposed that sulforaphane, a chemical found in broccoli, arugula, bok choy, kale, brussels sprouts, and other green vegetables, can help set the Nrf2 pathway into motion. Animal studies suggest that this nutrient can reduce oxidative stress and may be protective against certain diabetes complications, but this research is still ongoing. Curcumin, which is found in turmeric, has also been shown to be an Nrf2 activator.
Research suggests that a group of compounds called alkyl-catechols, which are found in fermented foods, may also activate this master pathway. As one 2016 paper explains, these three compounds (4-vinylcatechol, 4-ethylcatechol, and 4-methylcatechol) were once prevalent in ancient diets. Today’s western diets, the study notes, often lack these compounds. But early evidence suggests that fermented foods still around today, like kimchi, can activate this pathway and stimulate the antioxidant defense system.
Certain foods may also stoke the body’s ability to produce specific antioxidants. One of the primary antioxidants produced by the body is glutathione, a potent antioxidant that can bind up ROS and may help repair DNA. One 2019 paper suggested that foods like green tea, lean proteins, salmon, cruciferous vegetables, and turmeric may support glutathione production.
Some studies indicate that sulfur-rich foods support glutathione production because a vital part of the molecule itself is sulfur. Some sulfuric foods proposed to help support glutathione production include asparagus, avocado, cucumber, green beans, spinach, cruciferous vegetables, as well as alliums like onions or garlic.
Dietary antioxidants
So-called dietary antioxidants aren’t made by the body but can have similar effects when consumed in food. They’re especially plentiful in fruits and vegetables. Vitamin C, vitamin E, carotenoids (found in yellow and orange vegetables like squash and carrots), and polyphenols (found in berries, kiwis, plums, cherries, and apples) are some examples of dietary antioxidants.
Diets rich in plant-based food are protective against oxidative stress. For example, one study on 54 people with Type 2 diabetes examined how fruit and vegetable intake was related to antioxidant levels (and, by extension, protection against oxidative stress). It found strong links between dietary antioxidant levels and fruit and vegetable intake, which included vegetables, root crops, fruits, berries, and jams and preserves made from these foods. Specifically, the antioxidants vitamin C, plasma γ-tocopherol, and ascorbate were negatively correlated to oxidative stress. Overall, people who ate more fruit and vegetables also showed fewer signs of oxidative stress.
However, the full impact of dietary antioxidants—especially supplements—is not fully understood. Specific vitamins are known to support the antioxidant system still show inconsistent results in preventing the consequences of oxidative stress. For example, while observational studies have shown that when people eat more antioxidants (like vitamin C), they tend to have lower risks of coronary heart disease, clinical trials have demonstrated lackluster results. One meta-review published in 2019 of 10 of these trials found only weak evidence that vitamin C supplements lowered the risk of cardiovascular disease. As a partial explanation, the authors pointed to the lack of good-quality studies on vitamin C supplementation (most of those included in the review had small sample sizes) and noted that previous findings showing the benefits of vitamin C supplements may have resulted from “residual confounding and reverse causation.”
That doesn’t mean dietary antioxidants don’t have a role in fighting disease. But there’s much more to learn about how to stoke the body’s antioxidant defenses to ultimately prevent damage caused by oxidative stress.
Habits that support the antioxidant system
Behavior and habits also play a role in maintaining healthy levels of antioxidants and preventing the onset of oxidative stress.
Getting enough sleep is one example. Multiple animal studies have shown that sleep loss can raise levels of ROS in the brain, though the damage seems to show up elsewhere in the body, like the gut. In one 2020 study, researchers suggested that fruit flies eventually died from sleep deprivation partially because of ROS accumulation in their guts.
But like so many other parts of the oxidative stress story, sleep and ROS don’t have a simple hero-villain relationship. Sometimes, ROS play a key role in telling the body that it needs to get more sleep.
One 2018 study on fruit flies found that animals with chronic sleep loss tend to accumulate more ROS in brain tissue, and those high ROS levels reduced sleep. But sleep itself acted as an antioxidant, helping to clear out particular ROS. The decline of those ROS levels during sleep sent a biological signal that promoted wakefulness, resetting the cycle.
Taken together, regular sleep seems to be one way to keep ROS and antioxidants in balance, and ROS signaling plays a role in controlling sleep as well.
Exercise is another way to mediate oxidative stress and demonstrates that ROS can be helpful at certain levels and damaging at high levels. Though exercise can boost ROS production in the short term, it also seems to stimulate the antioxidant system. A recent study found that NADPH oxides (known as NOX)—oxygen transport proteins—are generated by exercise and help create ROS. Still, they also lead to adaptive processes that ultimately reduce oxidative stress and insulin resistance. One study on 34 men found higher levels of antioxidants after a single cycling session regardless of whether they put in the maximum or moderate effort. Over time, however, training seems to strengthen antioxidant responses. A small study on 16 women with obesity who followed a six-month exercise program (one hour of exercise, three times per week) found that they had increased levels of key antioxidant enzymes, even when the women were at rest.
Oxidative stress can seem like an inevitable consequence of living. But by eating well, maintaining healthy habits, and avoiding some known ROS stimulators, it’s possible to keep the ROS-antioxidant balance in check.








